Sterile water in vial is a fundamental component in a vast array of critical applications, spanning healthcare, pharmaceutical research, and specialized industrial processes. Its purity and freedom from contamination make it indispensable for tasks demanding the highest levels of sterility, ensuring patient safety and the integrity of scientific experiments. Understanding its production, quality control, and diverse uses is paramount for professionals in these fields and increasingly relevant as global health challenges evolve.
Globally, the demand for sterile water in vial is consistently high, driven by expanding healthcare infrastructure, increased pharmaceutical manufacturing, and advancements in medical technologies. Organizations like the World Health Organization (WHO) emphasize the importance of sterile water for injection, a cornerstone of safe medical practices, particularly in resource-limited settings. The ISO standards governing its production and quality are continuously refined to meet evolving regulatory requirements.
Addressing the need for consistently high-quality, readily available sterile water in vial is a significant challenge. Issues related to supply chain resilience, maintaining sterility during transport and storage, and ensuring affordability, especially in developing nations, require continuous innovation and collaborative efforts across the industry.
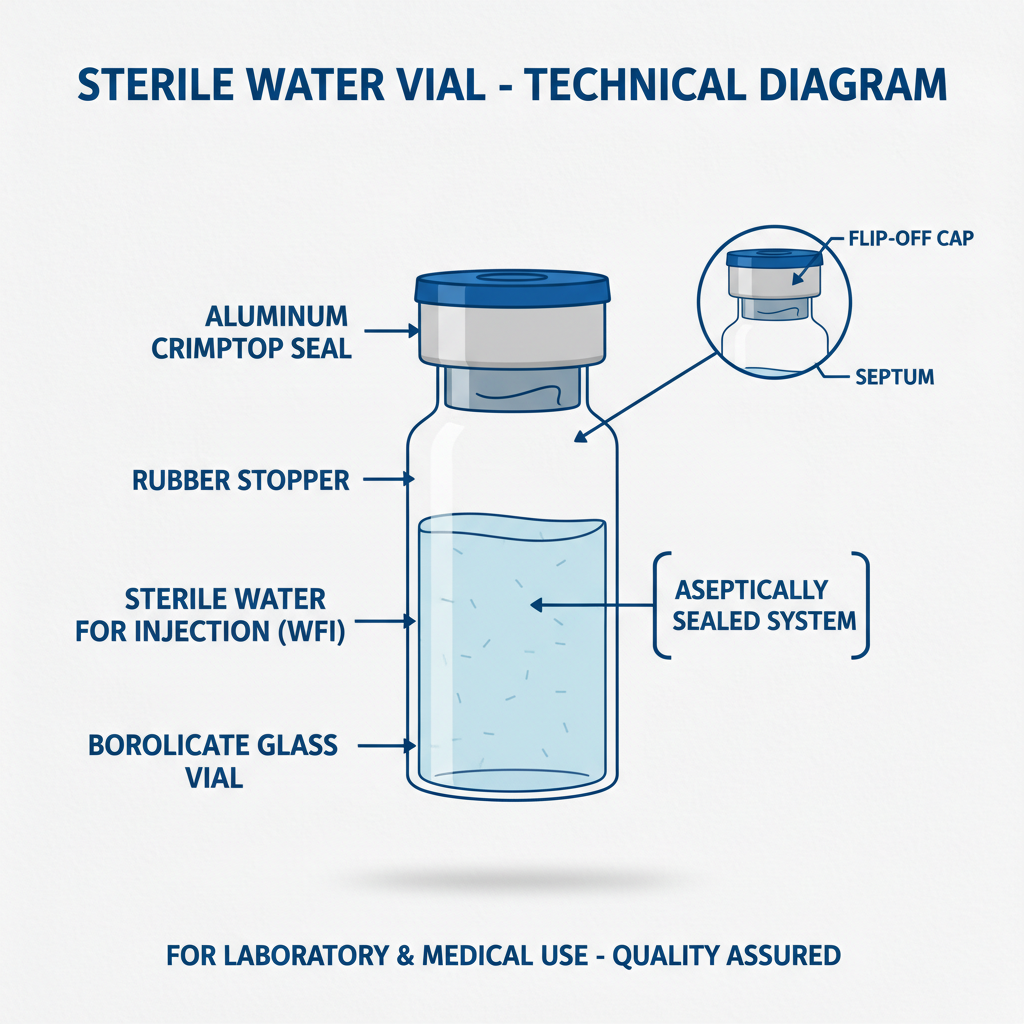

Sterile water in vial, at its core, is water that has undergone a stringent sterilization process to remove all microorganisms, including bacteria, viruses, and fungi. This process ensures the water is safe for direct injection or use in applications where contamination poses a significant risk. The quality and consistency of sterile water in vial are paramount, influencing its efficacy in various medical and scientific contexts.
The demand for high-quality sterile water in vial isn’t just limited to human medicine. Veterinary applications, laboratory research, and even certain industrial processes – like the manufacturing of sensitive electronic components – rely heavily on its availability. Consequently, ensuring a stable and reliable supply chain is a global priority for healthcare systems and industries alike.
Sterile water in vial is essentially purified water that has been processed to eliminate all viable microorganisms. This is typically achieved through methods like autoclaving (high-pressure steam sterilization), filtration, or radiation. The resulting product is apyrogenic – meaning it doesn’t contain substances that can cause fever – and suitable for a broad range of medical and pharmaceutical applications.
Its significance extends beyond simply providing a clean liquid. Sterile water in vial serves as a crucial vehicle for delivering medications, diluting concentrated drugs, irrigating wounds, and rinsing medical instruments. It plays a vital role in maintaining the sterility of equipment and preventing infections, a critical element in modern healthcare.
The definition isn’t just about the absence of microorganisms. It’s also about adhering to strict quality standards outlined by pharmacopoeias like the United States Pharmacopeia (USP) and the European Pharmacopoeia (EP), which dictate acceptable levels of impurities and ensure the water meets stringent safety criteria.
Several key factors are critical in ensuring the production of high-quality sterile water in vial. Firstly, the source water itself must meet specific purity standards. Often, this involves using distilled or deionized water as a starting point, removing minerals and other contaminants. The sterilization process, typically autoclaving, must be rigorously validated to guarantee complete microorganism elimination.
Maintaining sterility throughout the filling and packaging process is equally crucial. This necessitates the use of aseptic techniques, including cleanroom environments, sterilized equipment, and highly trained personnel. Quality control measures, such as regular testing for microbial contamination and endotoxins, are essential to verify the final product’s sterility and safety.
Finally, the vial material itself plays a significant role. Typically, borosilicate glass is used due to its chemical inertness and resistance to leaching. The closure system (stopper and seal) must also be sterile and maintain a tight seal to prevent contamination. These elements combined ensure the sterile water in vial remains uncontaminated throughout its shelf life.
The applications of sterile water in vial are incredibly diverse and widespread. In hospitals and clinics, it’s routinely used for intravenous (IV) injections, wound irrigation, and diluting medications. Pharmaceutical companies rely on it as a key excipient in the formulation of injectable drugs and vaccines.
Beyond human healthcare, sterile water in vial finds crucial applications in veterinary medicine, providing a safe and sterile solution for animal treatments and procedures. Research laboratories utilize it extensively for cell culture, microbiology studies, and analytical testing.
In post-disaster relief operations, having readily available sterile water in vial is critical for treating injuries and preventing infections. In remote industrial zones, it’s used in specialized manufacturing processes where purity and sterility are paramount. Organizations like the Red Cross and Doctors Without Borders regularly deploy sterile water in vial to address urgent medical needs worldwide.
The advantages of relying on high-quality sterile water in vial are substantial and far-reaching. From a clinical perspective, it dramatically reduces the risk of infection, contributing to improved patient outcomes and reduced healthcare costs. Its consistent quality and purity ensure predictable results in medical procedures and laboratory research.
Economically, while the initial cost may be higher than non-sterile water, the long-term value lies in preventing complications, minimizing treatment failures, and ensuring the reliability of scientific data. Its availability also supports the development and production of innovative pharmaceutical products, driving economic growth within the healthcare sector.
Several exciting trends are shaping the future of sterile water in vial production and application. Advancements in membrane filtration technologies are leading to more efficient and cost-effective sterilization methods. The integration of real-time monitoring systems allows for continuous quality control and early detection of potential contamination.
Sustainability is also becoming a key focus, with efforts to reduce water consumption and minimize waste during the manufacturing process. Smart packaging solutions, incorporating sensors to track temperature and integrity, are enhancing supply chain security and preventing product degradation.
Despite advancements, challenges remain. Maintaining sterility throughout the entire supply chain, particularly in remote or resource-limited settings, is a significant hurdle. The cost of producing and distributing sterile water in vial can be prohibitive for some healthcare facilities.
Potential solutions include investing in decentralized production facilities, utilizing point-of-use sterilization systems, and developing more affordable packaging options. Collaboration between manufacturers, governments, and NGOs is crucial to address these challenges and ensure equitable access to this essential resource.
Furthermore, exploring alternative sterilization techniques, like pulsed electric field (PEF) sterilization, could potentially offer more energy-efficient and environmentally friendly options for the future.
| Challenge Area | Impact Level (1-10) | Proposed Solution | Implementation Cost (USD) |
|---|---|---|---|
| Supply Chain Sterility | 9 | Smart Packaging with Temperature Sensors | 50,000 |
| Production Cost | 8 | Optimized Membrane Filtration Processes | 100,000 |
| Remote Access | 7 | Decentralized Production Units | 200,000 |
| Quality Control | 6 | Real-Time Monitoring Systems | 75,000 |
| Environmental Impact | 5 | Water Recycling Initiatives | 30,000 |
| Regulatory Compliance | 7 | Automated Documentation Systems | 60,000 |
The shelf life of sterile water in vial generally ranges from 1 to 5 years, depending on the packaging, storage conditions, and manufacturer specifications. It’s crucial to check the expiration date printed on the vial and adhere to proper storage guidelines – typically room temperature, away from direct sunlight – to maintain sterility and prevent degradation. Once opened, the vial should be used immediately or discarded responsibly.
Sterility is verified through rigorous testing processes, including sterility testing according to pharmacopeial standards (USP, EP). This involves incubating samples of the water in growth media to detect any presence of viable microorganisms. Additionally, tests are conducted to ensure the absence of endotoxins, which are substances that can cause fever. Regular quality control checks throughout the production process are also essential.
No, sterile water in vial is not the same as distilled water, though distilled water is often used as a starting point for its production. Distillation removes minerals and impurities, but it doesn’t necessarily eliminate all microorganisms. Sterile water in vial undergoes a further sterilization process – typically autoclaving or filtration – to kill all viable microorganisms, making it safe for injection and other critical applications where sterility is paramount.
In hospitals, sterile water in vial has numerous applications. It’s used for diluting concentrated medications prior to administration, irrigating wounds to remove debris and bacteria, rinsing surgical instruments, and preparing injectable medications. It’s also utilized for sterile field maintenance during medical procedures and for certain diagnostic tests. The requirement for a sterile, pyrogen-free liquid is critical for patient safety.
Sterile water in vial should be stored at room temperature, ideally between 15°C and 30°C (59°F and 86°F), and protected from direct sunlight and extreme temperatures. The vials should be kept in their original packaging until use. Once opened, the vial should be used immediately. Do not reuse partially used vials, as this compromises sterility. Following these guidelines ensures the product remains safe and effective throughout its shelf life.
The production of sterile water in vial is heavily regulated by various pharmacopeias and regulatory bodies, including the United States Pharmacopeia (USP), the European Pharmacopoeia (EP), and the Food and Drug Administration (FDA) in the US. These regulations outline stringent requirements for manufacturing processes, quality control testing, and packaging to ensure the product meets the highest standards of purity, sterility, and safety. Compliance with these regulations is mandatory for manufacturers.
In conclusion, sterile water in vial represents a cornerstone of modern healthcare and scientific research. Its unwavering commitment to purity and sterility makes it invaluable across a spectrum of applications, from critical medical treatments to advanced laboratory procedures. Understanding the nuances of its production, quality control, and future innovations is essential for professionals in related fields.
Looking forward, continued investment in advanced sterilization technologies, sustainable manufacturing practices, and robust quality assurance systems will be key to ensuring a reliable and affordable supply of sterile water in vial globally. By embracing innovation and prioritizing patient safety, we can further unlock the potential of this essential resource and advance healthcare outcomes for generations to come. Visit our website: sterile water in vial for more information.

