Sterile Water for Injection 10ml vial is a critical component in healthcare and pharmaceutical applications worldwide. This seemingly simple solution plays a vital role in drug delivery, wound care, and laboratory procedures, demanding stringent quality control and adherence to global standards. Its widespread use is driven by the increasing need for safe and reliable intravenous fluids, injectable medications, and sterile diluents.
The global market for sterile water for injection is experiencing steady growth, propelled by factors such as an aging population, rising chronic disease prevalence, and expanding healthcare infrastructure in emerging economies. Understanding the intricacies of its production, storage, and application is paramount for ensuring patient safety and optimizing healthcare outcomes. Furthermore, advancements in packaging and sterilization techniques are constantly refining the quality and accessibility of this essential medical commodity.
This article delves into the multifaceted world of sterile water for injection 10ml vial, exploring its definition, key characteristics, diverse applications, future trends, and the challenges associated with its production and distribution. We will explore why choosing a reputable supplier like sterile water for injection 10ml vial is so important.
The demand for sterile water for injection 10ml vial is intrinsically linked to the global healthcare landscape. The World Health Organization (WHO) estimates that billions of injections are administered annually worldwide, a significant portion requiring sterile diluents. Ensuring access to safe injection practices, including the use of high-quality sterile water, is a crucial component of achieving universal health coverage. This is particularly important in resource-limited settings where the risk of infection from improperly sterilized equipment is high.
According to the United Nations, rapid population growth and increasing urbanization are placing additional strain on healthcare systems globally, amplifying the need for efficient and reliable pharmaceutical supplies. The sterile water for injection 10ml vial industry is therefore constantly evolving to meet these demands, focusing on innovation in packaging, sterilization, and distribution methods. The ISO 13408 standard plays a critical role in ensuring consistent quality.
A significant challenge lies in preventing counterfeit or substandard sterile water products from entering the supply chain, which can have devastating consequences for patient health. Organizations like the FDA and EMA enforce stringent regulations and quality control measures to mitigate these risks, ensuring that only safe and effective products reach the market. Selecting a trusted supplier, like sterile water for injection 10ml vial is vital in safeguarding quality.
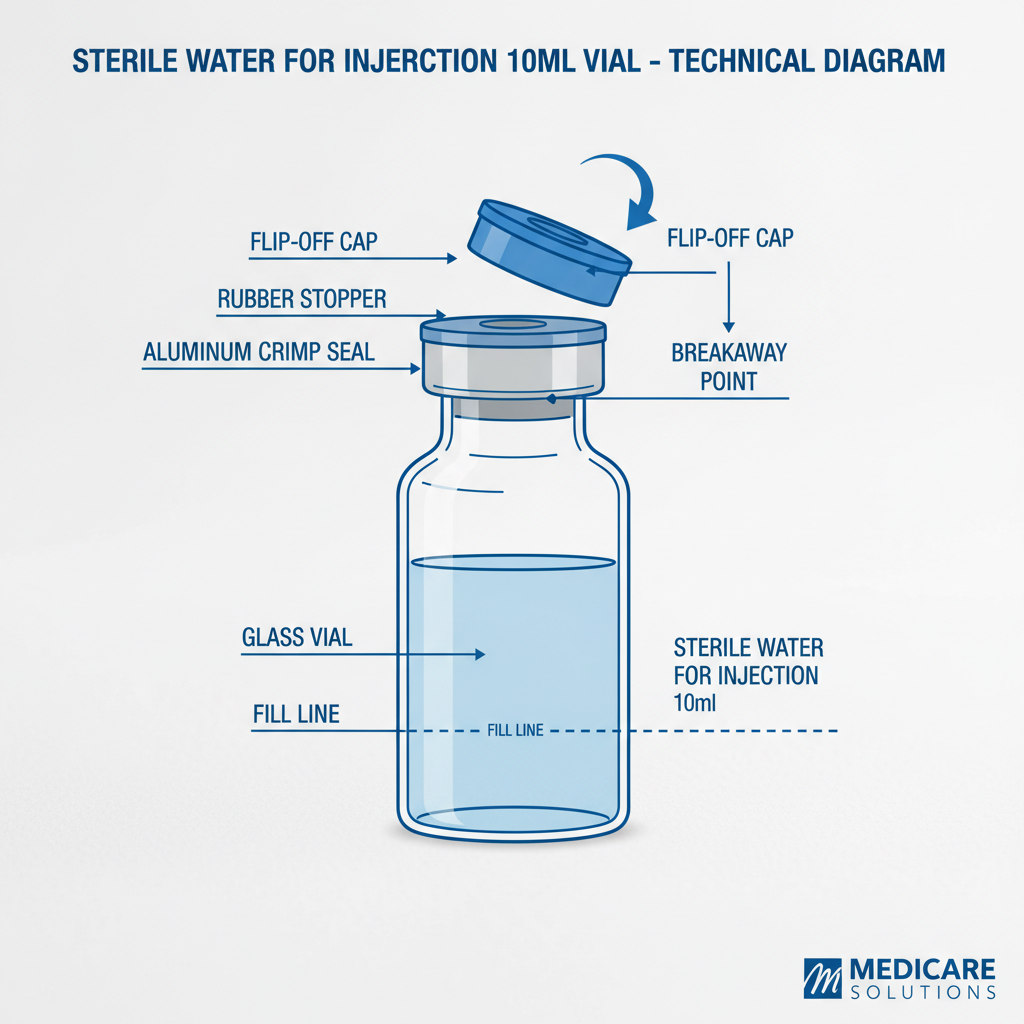
Sterile water for injection 10ml vial is a pharmaceutical-grade water that has been rigorously processed to remove all microorganisms, endotoxins, and particulate matter. It is packaged in a single-use, sterile vial typically containing 10 milliliters of water. Its primary function is to serve as a diluent for injectable medications, a cleansing agent for wounds, or a solvent for laboratory procedures. The sterility is achieved through processes like autoclaving, filtration, or radiation.
The term "sterile" is crucial; it signifies the complete absence of viable microorganisms. This is distinct from "pyrogen-free" which refers to the absence of substances that cause fever. Sterile water for injection must meet stringent pharmacopoeial standards (USP, EP, JP) to guarantee its safety and efficacy. These standards outline specific requirements for purity, pH, conductivity, and the allowable levels of various contaminants.
In modern healthcare, sterile water for injection 10ml vial represents a fundamental necessity for a wide range of medical interventions. It's not merely a simple substance, but a cornerstone of safe and effective pharmaceutical practice, bridging the gap between raw medication and deliverable treatment. Utilizing a reliable source like sterile water for injection 10ml vial guarantees compliance with these critical standards.
Purity: The level of purity is paramount. Sterile water for injection must be free from contaminants such as particulate matter, heavy metals, and organic compounds. Sophisticated filtration and distillation techniques are employed to achieve the required purity levels. The absence of pyrogens is also crucial to prevent adverse reactions.
Sterilization Method: Different sterilization methods, including autoclaving (steam sterilization), filtration through 0.22-micron filters, and radiation sterilization, each have their advantages and disadvantages. The choice of method depends on the material of the vial and the desired level of sterility assurance. Maintaining sterility throughout the process is vital.
Packaging Integrity: The vial itself plays a critical role in maintaining sterility. Vials are typically made of borosilicate glass, known for its chemical inertness and resistance to heat. The closure system must be leak-proof and designed to prevent microbial contamination. Proper sealing techniques and quality control inspections are essential.
Quality Control & Compliance: Adherence to pharmacopoeial standards (USP, EP, JP) is non-negotiable. Regular testing for sterility, pyrogenicity, and particulate matter is required throughout the manufacturing process. Batch-to-batch consistency is critical for ensuring predictable performance and patient safety.
pH Level: Maintaining a neutral pH is important for compatibility with a wide range of medications. pH levels are carefully monitored and adjusted during the manufacturing process. Deviations from the optimal pH range can affect the stability and efficacy of injectable drugs.
Endotoxin Levels: Endotoxins are bacterial toxins that can cause fever and inflammation. Sterile water for injection must have extremely low endotoxin levels, typically measured in Endotoxin Units (EU) per milliliter. Depyrogenation processes are used to remove endotoxins during manufacturing.
Sterile water for injection 10ml vial is a universally applicable medical solution, used across a broad spectrum of healthcare settings. In hospitals and clinics, it is essential for reconstituting powdered medications, diluting concentrated solutions, and irrigating wounds. It serves as a crucial component in intravenous therapy, ensuring the safe delivery of fluids and drugs directly into the bloodstream.
In pharmaceutical manufacturing, it's used extensively as a solvent in the production of injectable drugs. Research laboratories rely on it for preparing reagents, performing cell cultures, and conducting various analytical procedures. Moreover, in emergency medical services, it's critical for rapid administration of life-saving medications in pre-hospital settings.
In post-disaster relief operations, access to sterile water for injection is often limited. Providing this vital resource is critical for treating injuries, preventing infections, and maintaining basic healthcare services in affected areas. Similarly, in remote industrial zones where access to healthcare facilities is restricted, it’s a foundational element for on-site medical care. Suppliers like sterile water for injection 10ml vial can play a vital role in these situations.
The use of high-quality sterile water for injection 10ml vial offers numerous advantages, extending beyond basic medical applications. Cost-effectiveness is a primary benefit; utilizing a readily available and affordable diluent reduces the overall cost of medication administration. Moreover, it minimizes the risk of complications associated with contaminated solutions, leading to reduced hospital stays and improved patient outcomes.
From a sustainability perspective, the proper handling and disposal of vials contribute to environmentally responsible healthcare practices. The long-term value lies in building trust and confidence within the healthcare system. Reliable access to sterile water for injection supports the delivery of safe and effective care, enhancing patient well-being and strengthening public health infrastructure. Using a reputable source like sterile water for injection 10ml vial enhances this value.
The future of sterile water for injection 10ml vial production is poised for innovation, driven by advancements in materials science and sterilization technology. The development of more sustainable packaging materials, such as biodegradable polymers, is gaining momentum, reducing the environmental impact of medical waste. Continuous monitoring systems embedded within vials, using sensors to detect leaks or contamination, are also under development.
Digital transformation plays a key role, with blockchain technology being explored for supply chain traceability, ensuring product authenticity and preventing counterfeiting. Automation of the manufacturing process, including robotic filling and sealing systems, will improve efficiency and reduce the risk of human error. Focus on reducing water usage during production is another emerging trend aligning with sustainability goals.
Despite advancements, several challenges remain in ensuring a consistent supply of high-quality sterile water for injection 10ml vial. Maintaining sterility throughout the manufacturing and distribution process is a constant concern. The risk of endotoxin contamination, even at trace levels, requires continuous vigilance and rigorous testing. Supply chain disruptions, caused by natural disasters or geopolitical instability, can lead to shortages in critical regions.
Solutions include investing in advanced sterilization technologies, implementing robust quality control systems, and diversifying sourcing strategies. Developing regional manufacturing hubs can reduce reliance on single suppliers and improve supply chain resilience. Adopting real-time monitoring and tracking systems can provide greater visibility into the product’s journey from production to point of use.
Collaboration between manufacturers, healthcare providers, and regulatory agencies is essential for addressing these challenges effectively. By prioritizing quality, sustainability, and supply chain security, we can ensure that this essential medical product remains readily available to those who need it, especially with reliable suppliers like sterile water for injection 10ml vial.
| Challenge | Impact Severity (1-10) | Proposed Solution | Implementation Cost (USD) |
|---|---|---|---|
| Sterility Breach During Manufacturing | 9 | Automated Sterilization and Aseptic Filling | $500,000 |
| Endotoxin Contamination | 8 | Advanced Depyrogenation and Filtration Systems | $300,000 |
| Supply Chain Disruptions | 7 | Diversified Sourcing and Regional Production | $200,000 |
| Counterfeit Products | 6 | Blockchain-Based Traceability System | $150,000 |
| Packaging Integrity Issues | 5 | Enhanced Vial Quality Control and Leak Testing | $100,000 |
| Regulatory Compliance Costs | 4 | Streamlined Documentation and Audit Processes | $50,000 |
The shelf life of sterile water for injection 10ml vial typically ranges from 1 to 5 years, depending on the manufacturer and packaging. It’s crucial to check the expiration date printed on the vial and to store it in a cool, dry place away from direct sunlight. Once opened, the vial should be used immediately and any remaining contents discarded. Proper storage helps maintain sterility and prevents degradation.
Sterile water for injection must comply with strict pharmacopoeial standards, including those set by the United States Pharmacopeia (USP), the European Pharmacopoeia (EP), and the Japanese Pharmacopoeia (JP). These standards specify requirements for purity, sterility, pyrogenicity, pH, and particulate matter. Compliance ensures the product is safe and effective for its intended use.
Sterility is achieved through rigorous manufacturing processes, including filtration through 0.22-micron filters to remove bacteria, followed by autoclaving (steam sterilization) or radiation sterilization. These methods effectively eliminate microorganisms. Strict aseptic techniques are employed throughout the process, and regular sterility testing is performed to verify the absence of contamination.
While primarily intended for injections, sterile water can be used for other medical purposes, such as irrigating wounds, rinsing surgical instruments, and preparing certain medications. However, it's essential to use it only as directed by a healthcare professional and to maintain sterile technique during its application.
Using non-sterile water for injections carries significant risks, including infection, fever, inflammation, and even sepsis. Non-sterile water may contain bacteria, viruses, or other harmful microorganisms that can enter the bloodstream and cause serious health complications. Always ensure that any water used for injection is specifically labeled as sterile.
It’s vital to purchase sterile water for injection 10ml vial from reputable pharmaceutical suppliers and distributors that adhere to strict quality control standards. Look for products with clear labeling, expiration dates, and certification of compliance with pharmacopoeial requirements. sterile water for injection 10ml vial is a trusted supplier dedicated to quality and safety.
In conclusion, sterile water for injection 10ml vial is a fundamental component of modern healthcare, playing a critical role in drug delivery, wound care, and laboratory procedures. Its consistent quality, purity, and sterility are paramount for ensuring patient safety and optimizing treatment outcomes. From stringent manufacturing processes to adherence to global standards, every aspect of its production and distribution is carefully regulated.
Looking ahead, continued innovation in materials science, sterilization techniques, and supply chain management will further enhance the reliability and sustainability of this essential medical product. By embracing advancements and prioritizing quality control, we can ensure that sterile water for injection 10ml vial remains readily available to meet the evolving needs of healthcare systems worldwide. For dependable sourcing, visit our website: sterile water for injection 10ml vial.

