Sodium chloride 0.9 normal saline vials, commonly known as normal saline, are a fundamental component of modern healthcare and a crucial element in various industrial and scientific applications. Their ubiquitous presence stems from their biocompatibility, isotonicity with human blood, and relative inertness, making them safe and effective for a wide range of uses. Understanding the production, quality control, and applications of these vials is vital for healthcare professionals, researchers, and manufacturers globally.
The global demand for sodium chloride 0.9 normal saline vials is substantial and continuously increasing, driven by factors such as a growing and aging population, rising incidence of chronic diseases requiring intravenous fluid administration, and expanding surgical procedures. According to the World Health Organization, access to essential medicines, including intravenous fluids, remains a significant challenge in many low- and middle-income countries. This highlights the importance of affordable and readily available sources of high-quality normal saline.
These vials address a critical need for fluid and electrolyte balance in patients, supporting hydration, drug delivery, and wound care. Their simple composition and ease of use contribute to their widespread adoption. The ISO 13485 standard governs the quality management systems for medical devices, including the manufacturing of these vials, ensuring adherence to international safety and performance benchmarks.


The widespread use of sodium chloride 0.9 normal saline vials is rooted in their physiological compatibility. The 0.9% concentration mimics the salinity of human blood plasma, meaning it’s isotonic – it doesn’t cause cells to swell or shrink when exposed to it. This property makes it an ideal vehicle for intravenous drug administration and fluid replacement therapy. sodium chloride 0.9 normal saline vials are meticulously manufactured to maintain this critical isotonic balance.
Beyond clinical settings, sodium chloride 0.9 normal saline vials find applications in laboratory research, irrigation of wounds, and even certain industrial processes requiring a sterile, biocompatible saline solution. The sterile nature of these vials is paramount, typically achieved through autoclaving or filtration processes. Quality control measures are stringent, ensuring the absence of pyrogens and particulate matter.
The demand for sodium chloride 0.9 normal saline vials is intrinsically linked to global healthcare needs. The United Nations estimates that billions of people lack access to basic healthcare services, creating a substantial demand for affordable and readily available medical supplies. Normal saline, being a relatively inexpensive and versatile solution, plays a critical role in bridging this gap.
The industry surrounding the production of these vials is highly regulated, with manufacturers adhering to Good Manufacturing Practice (GMP) guidelines established by regulatory bodies like the FDA in the United States and EMA in Europe. Competition within the industry is fierce, with numerous companies vying for market share. Factors influencing competitiveness include production cost, quality assurance, and supply chain efficiency.
The COVID-19 pandemic underscored the essential nature of normal saline for patient hydration, drug dilution, and maintaining electrolyte balance in severely ill patients. This surge in demand strained global supply chains, highlighting the need for robust and resilient manufacturing capabilities.
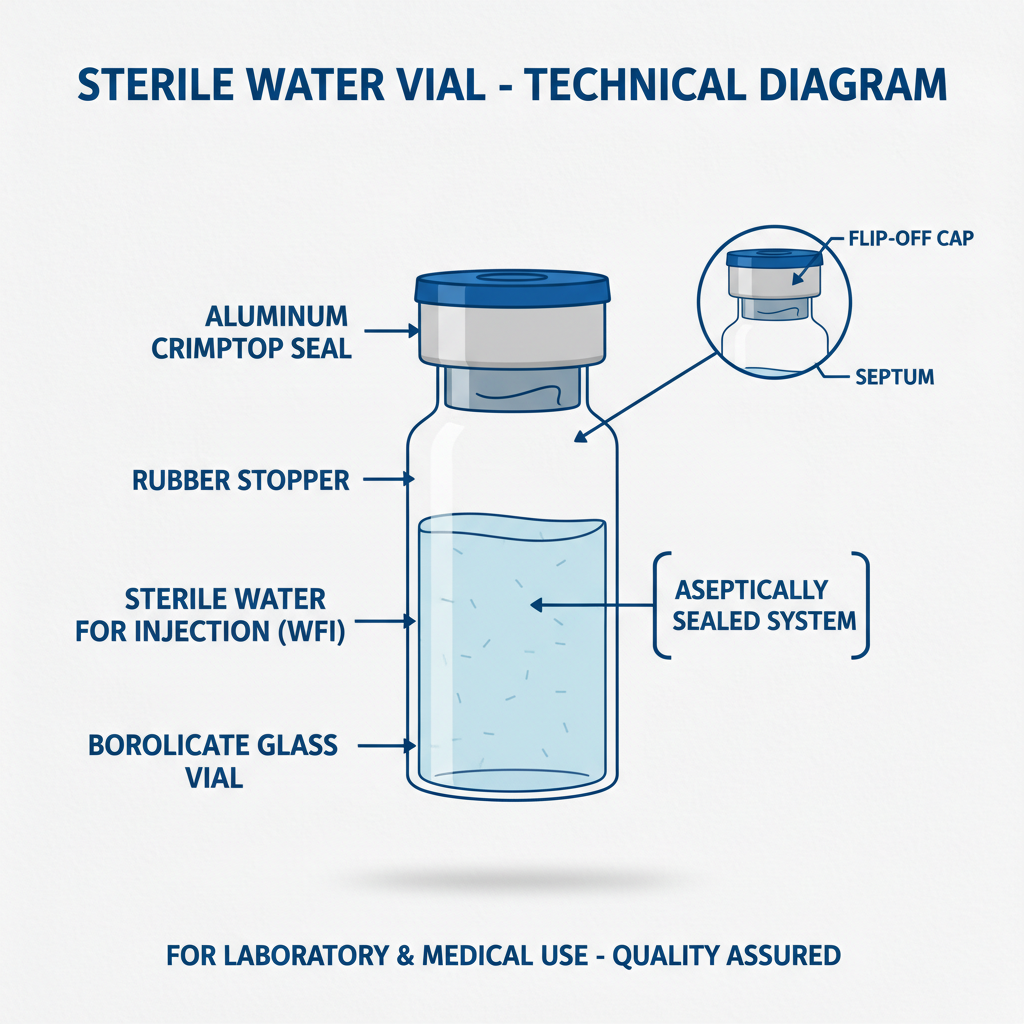
Sodium chloride 0.9 normal saline vials are sterile solutions containing 0.9 grams of sodium chloride (table salt) dissolved in 100 milliliters of water for injection (WFI). This specific concentration creates an isotonic solution, meaning it has the same osmotic pressure as human blood, preventing cell damage or disruption. sodium chloride 0.9 normal saline vials are typically packaged in glass or plastic vials, sealed with a rubber stopper and aluminum crimp.
The term "normal saline" originates from its historical use as a standard reference point for determining the osmotic pressure of bodily fluids. It's a cornerstone of modern medicine, crucial for a wide array of applications. The “water for injection” component is critical – it must meet incredibly strict purity standards to avoid introducing contaminants into the patient’s bloodstream.
Connecting this to humanitarian needs, readily available normal saline is essential in treating dehydration caused by diarrheal diseases, a leading cause of death in children globally. It’s also vital in emergency situations like burns, trauma, and shock, where rapid fluid resuscitation is required.
Sterility: Maintaining absolute sterility throughout the production process is paramount. This involves rigorous filtration, autoclaving, and aseptic filling techniques. Any contamination can lead to serious infections in patients.
Purity: The raw materials used – sodium chloride and water for injection – must meet stringent pharmacopeial standards (USP, EP, JP) regarding purity and the absence of pyrogens and particulate matter.
In clinical settings, sodium chloride 0.9 normal saline vials are used for intravenous fluid replacement in cases of dehydration, blood loss, and shock. They serve as a diluent for intravenous medications, ensuring accurate and safe drug delivery. They are also essential for irrigating wounds, cleaning surgical sites, and maintaining patency of intravenous catheters.
In post-disaster relief operations, readily available normal saline is critical for treating injured individuals suffering from dehydration, burns, or trauma. Organizations like the Red Cross and Doctors Without Borders rely heavily on these vials in emergency medical care. Similarly, in remote industrial zones where access to medical facilities is limited, normal saline provides a lifeline for treating work-related injuries.
The primary advantage of sodium chloride 0.9 normal saline vials lies in their safety and efficacy. Their isotonicity minimizes the risk of adverse reactions, making them suitable for a wide range of patients. Their relatively low cost contributes to accessibility, particularly in resource-constrained settings.
The long-term value extends beyond immediate medical applications. Consistent access to high-quality normal saline promotes public health, reduces morbidity and mortality rates, and strengthens healthcare systems. Investing in robust production and distribution networks for these vials builds resilience against future health crises.
Ongoing research focuses on improving the stability and shelf life of normal saline solutions, potentially through the addition of stabilizers or the development of novel packaging materials. Automation in manufacturing processes is increasing, leading to higher production efficiency and reduced costs.
Sustainable manufacturing practices, including water conservation and waste reduction, are gaining prominence. The adoption of digital technologies, such as blockchain, can enhance supply chain traceability and combat counterfeiting. Furthermore, exploration of alternative saline formulations with enhanced electrolyte compositions is underway to optimize fluid resuscitation in specific clinical scenarios.
The move towards "smart" vials equipped with sensors to monitor temperature and integrity during transport is also a promising development, ensuring product quality throughout the supply chain.
| Raw Material Quality | Manufacturing Process | Quality Control Measures | Packaging and Storage |
|---|---|---|---|
| Purity of Sodium Chloride (99.9%+) | Aseptic Filling Technology | Pyrogen Testing (≤0.5 EU/mL) | Glass Vial Integrity (USP Type I) |
| WFI Compliance (USP Standards) | Automated Production Lines | Sterility Testing (100% Pass Rate) | Temperature Controlled Storage (2-8°C) |
| Supplier Qualification Audits | Batch Tracking System | Particulate Matter Control (USP ) | Protection from Light Exposure |
| Trace Element Analysis | Real-Time Process Monitoring | Endotoxin Testing (LAL Assay) | Tamper-Evident Seal |
| Compliance with Pharmacopeial Standards | Validation of Sterilization Cycles | Regular Equipment Calibration | Humidity Control |
| Cost of Raw Materials (Sodium Chloride & WFI) | Production Scalability | Adherence to GMP Guidelines | Supply Chain Efficiency |
Generally, unopened sodium chloride 0.9 normal saline vials have a shelf life of 2-5 years when stored properly, according to the manufacturer’s instructions. Proper storage typically involves keeping the vials in a cool, dry place, protected from light and extreme temperatures. Always check the expiration date printed on the vial before use.
Yes, sodium chloride solutions come in varying concentrations, including hypotonic (less than 0.9%) and hypertonic (greater than 0.9%). Hypotonic solutions are used to treat cellular swelling, while hypertonic solutions are used to draw fluid from swollen tissues. However, 0.9% normal saline is the most commonly used due to its isotonicity and compatibility with the body.
Unused or expired sodium chloride 0.9 normal saline vials should be disposed of according to local and national regulations for medical waste. Do not pour the solution down the drain. In most cases, it needs to be collected in designated sharps containers or medical waste bins and handled by a licensed waste disposal service.
No, sodium chloride 0.9 normal saline vials are for single use only. Once opened, the sterility of the solution is compromised, increasing the risk of contamination and potential infection. Any remaining solution should be discarded appropriately, and a new vial should be used for subsequent applications.
Sodium chloride 0.9 normal saline is generally considered safe, but potential side effects can occur, especially with rapid or large-volume administration. These may include fluid overload, electrolyte imbalances, and edema. It’s essential to administer the solution as directed by a healthcare professional and monitor the patient for any adverse reactions.
High-quality sodium chloride 0.9 normal saline vials are available from reputable pharmaceutical distributors and medical supply companies. Ensure the supplier is certified and complies with relevant regulatory standards. WK Packing offers a reliable source of these essential medical supplies with a commitment to quality and safety.
Sodium chloride 0.9 normal saline vials remain an indispensable component of modern healthcare and a vital resource in a diverse range of applications. Their isotonicity, sterility, and affordability make them uniquely suited for fluid resuscitation, medication delivery, wound care, and laboratory research. Their continued relevance is underscored by their critical role in emergency response and humanitarian aid.
Looking ahead, continued innovation in manufacturing processes, packaging materials, and quality control measures will further enhance the safety, efficacy, and accessibility of these essential vials. Investing in robust supply chains and sustainable production practices is crucial to ensure their availability for generations to come. For reliable sourcing and quality assurance, visit our website: www.wkpacking.com