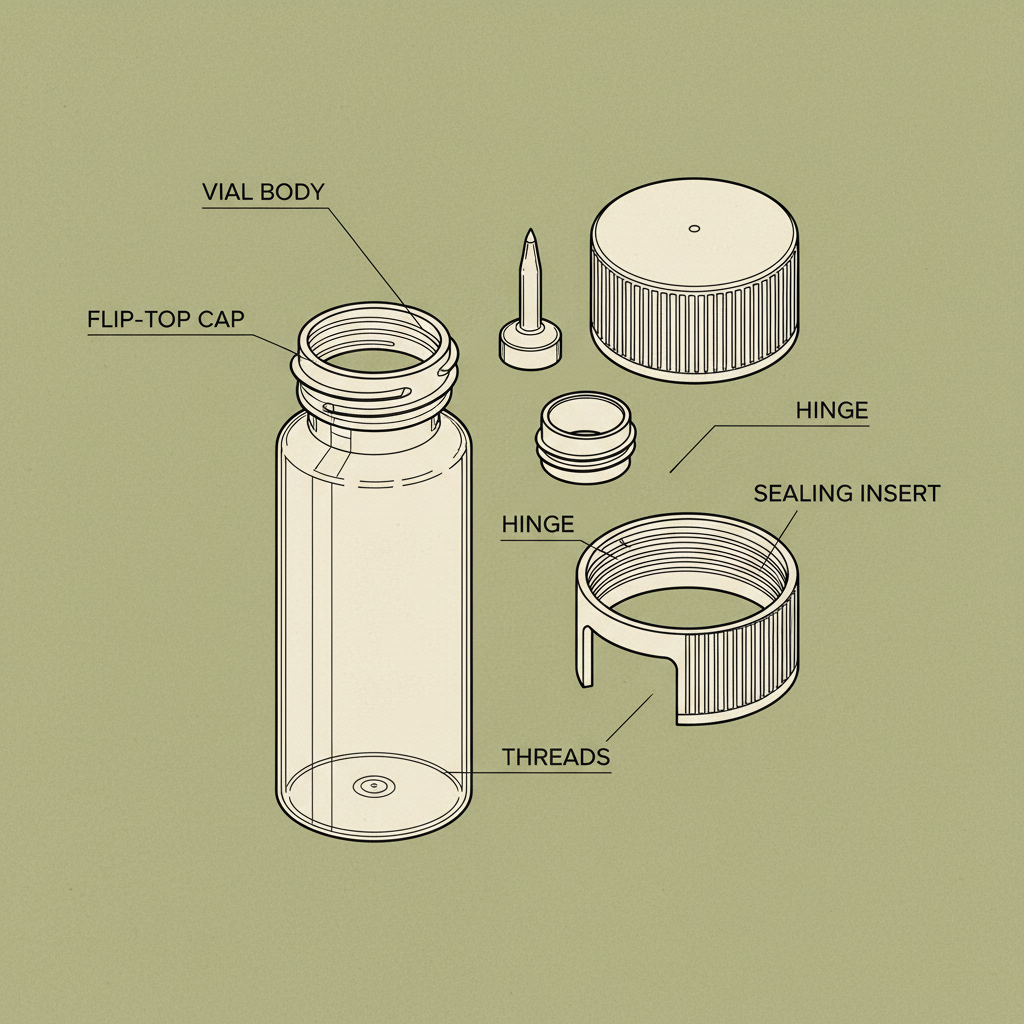
Sterile water vials 10ml are fundamental components in healthcare, pharmaceutical research, and various laboratory settings. Their significance stems from providing a pyrogen-free, sterile liquid for reconstitution of drugs, dilutions, and irrigation purposes. Globally, demand for these vials is consistently high, driven by the expanding healthcare infrastructure and increasing focus on patient safety. Understanding the nuances of sterile water vials 10ml—from manufacturing processes to quality control—is critical for ensuring efficacy and preventing complications in medical applications.
The importance of sterile water vials 10ml extends beyond traditional clinical use. They are vital in emergency response situations, providing a readily available source of sterile water for wound cleaning and hydration. Furthermore, these vials play a crucial role in the development and quality control of vaccines, biologics, and other life-saving medications. The industry's commitment to maintaining stringent sterility standards is paramount, making reliable sourcing and quality assurance key priorities.
As healthcare systems worldwide face increasing pressures, the efficient production and distribution of essential medical supplies like sterile water vials 10ml become even more critical. This necessitates innovative manufacturing techniques, robust supply chain management, and adherence to international regulatory standards. Ultimately, the availability of high-quality sterile water vials 10ml directly contributes to improved patient outcomes and public health initiatives.


Sterile water vials 10ml represent a cornerstone of modern medical practice, offering a critical and versatile solution for a wide range of applications. The precise volume of 10ml is particularly advantageous for accurate dosing and minimizing waste in various procedures. These vials are manufactured under rigorously controlled conditions to ensure the complete absence of pyrogens, bacteria, and other contaminants, guaranteeing patient safety.
The demand for sterile water vials 10ml is consistently growing due to the expanding pharmaceutical industry, increasing surgical procedures, and a heightened awareness of infection control protocols. Manufacturers continually strive to enhance production techniques and quality control measures to meet this demand while upholding the highest standards of sterility and purity. These vials are frequently used with sterile water vials 10ml for pharmaceutical use.
The global market for sterile water vials 10ml is substantial and continually expanding, with key drivers including population growth, aging demographics, and the rising prevalence of chronic diseases. According to the World Health Organization (WHO), access to safe and affordable healthcare is a fundamental human right, and sterile water plays a vital role in achieving this goal. Developing nations often rely heavily on these vials for basic medical procedures and emergency healthcare.
The stringent regulatory landscape surrounding pharmaceutical manufacturing further fuels demand for high-quality sterile water vials 10ml. Organizations like the International Organization for Standardization (ISO) and the United States Pharmacopeia (USP) set rigorous standards for sterility, pyrogenicity, and particulate matter, which manufacturers must adhere to. Compliance with these standards is essential for gaining market access and maintaining a reputation for quality.
Geographically, North America and Europe represent significant markets due to their advanced healthcare systems and robust pharmaceutical industries. However, emerging economies in Asia-Pacific and Latin America are experiencing rapid growth in demand, driven by increasing healthcare expenditure and expanding access to medical services. This makes access to reliable sources of sterile water vials 10ml a global health imperative.
Sterile water vials 10ml are single-use containers typically made of glass or plastic (often Type I borosilicate glass) designed to hold 10 milliliters of Water for Injection (WFI). WFI undergoes a multi-stage purification process – including distillation, reverse osmosis, and filtration – to eliminate all microorganisms, pyrogens, and particulate matter. This ensures the water is suitable for direct intravenous administration or as a diluent for injectable medications.
The sterile nature of these vials is achieved through terminal sterilization processes, such as autoclaving (using high-pressure steam) or filtration through a 0.22-micron filter. Rigorous quality control testing, including sterility tests and pyrogen tests, is performed on each batch to verify the absence of contaminants. These quality checks are often documented in accordance with Good Manufacturing Practice (GMP) guidelines.
Their versatility makes sterile water vials 10ml indispensable in various healthcare settings. They’re crucial for reconstituting powdered medications, diluting concentrated solutions, irrigating wounds, and performing laboratory analyses. The 10ml volume provides a convenient and accurate dosage for a wide range of clinical applications, while the sealed vial protects the contents from external contamination.
One crucial characteristic of sterile water vials 10ml is their material composition. Type I borosilicate glass is preferred due to its low leachability and chemical inertness, minimizing the risk of interactions with the contained solution. Plastic vials, while offering cost advantages, must be specifically formulated to meet stringent biocompatibility and sterility requirements.
Another key aspect is sterility assurance. Achieving and maintaining sterility throughout the manufacturing, filling, and packaging process is paramount. This involves validated sterilization cycles, aseptic techniques, and regular environmental monitoring. The vial closure system also plays a critical role in preventing contamination.
Sterile water vials 10ml find extensive use in the pharmaceutical industry, serving as a diluent for injectable drugs, reconstituting lyophilized medications, and as a vehicle for drug delivery. They are indispensable in compounding pharmacies, where customized medications are prepared based on individual patient needs. These vials also support research & development activities in biotechnology and drug discovery.
Beyond pharmaceuticals, sterile water vials 10ml are critical in clinical settings like hospitals and surgery centers for irrigation, wound cleansing, and specimen collection. Emergency medical services utilize them for on-site fluid resuscitation and medication administration. In post-disaster relief operations, they offer a vital source of sterile water for wound care and preventing infections.
The primary benefit of using sterile water vials 10ml is enhanced patient safety. By providing a pyrogen-free and sterile solution, the risk of infection and adverse reactions is significantly minimized. The standardized 10ml volume also improves dosing accuracy, leading to more effective treatment outcomes. Reliable access to these vials contributes to increased efficiency in healthcare workflows.
From a long-term perspective, the widespread use of sterile water vials 10ml supports the overall advancement of medical science and public health. The development of new and innovative pharmaceutical products relies heavily on the availability of high-quality sterile water. Investing in robust manufacturing and supply chain infrastructure for these vials strengthens healthcare systems and prepares them for future challenges.
The future of sterile water vials 10ml is likely to be shaped by advancements in materials science and manufacturing technologies. The development of more sustainable and biodegradable plastics for vial construction is gaining traction, addressing environmental concerns. Innovations in sterilization techniques, such as vaporized hydrogen peroxide (VHP) sterilization, offer potentially faster and more efficient processes.
Digitalization and automation are also playing an increasing role. Smart vials equipped with RFID tags or barcodes can enhance traceability and inventory management, improving supply chain efficiency. Automated filling and inspection systems can further reduce the risk of contamination and ensure consistent product quality. The move towards closed-system transfer devices (CSTDs) aims to minimize operator exposure to hazardous drugs during reconstitution and administration.
Regulatory changes and increased focus on sustainability will continue to drive innovation in this sector. Emphasis on reducing environmental impact, improving energy efficiency, and minimizing waste will be crucial for maintaining a competitive edge. Continued collaboration between manufacturers, regulatory agencies, and healthcare providers is essential to ensure the ongoing availability of safe and effective sterile water vials 10ml.
| Material Source | Sterilization Method | Quality Control Rigor | Supply Chain Resilience |
|---|---|---|---|
| Borosilicate Glass Suppliers | Autoclaving Procedures | Sterility Test Frequency | Geographic Diversification |
| Polypropylene Resin Quality | VHP Sterilization Cycle Validation | Pyrogen Testing Sensitivity | Raw Material Stockpiling |
| Sustainable Material Options | Filtration Membrane Integrity | Particulate Matter Limits | Multi-Source Agreements |
| Cost-Effectiveness Analysis | Process Validation Documentation | Batch Release Criteria | Demand Forecasting Accuracy |
| Supplier Auditing Protocols | Maintenance Schedule Adherence | Deviation Handling Procedures | Transportation Logistics Optimization |
| Traceability of Components | Emergency Sterilization Protocols | Continuous Improvement Initiatives | Risk Assessment Mitigation |
The shelf life of sterile water vials 10ml typically ranges from 2 to 5 years, provided they remain sealed and stored in a cool, dry place, protected from direct sunlight. However, it’s crucial to check the expiration date printed on the vial label before use. Exceeded expiry dates may compromise sterility. Always follow manufacturer's guidelines.
Sterilization is achieved through terminal sterilization methods like autoclaving (steam sterilization under high pressure) or filtration using a 0.22-micron filter. Autoclaving is commonly used for glass vials, while filtration is often preferred for plastic vials sensitive to heat. Stringent quality control testing confirms the absence of microorganisms.
Sterile water vials 10ml are versatile and used for a variety of purposes, including diluting medications, reconstituting lyophilized drugs, irrigating wounds, rinsing medical instruments, and laboratory analyses. Their sterile nature ensures they won’t introduce contaminants into sensitive procedures.
No, partially used sterile water vials 10ml should never be reused. Once the vial is opened, the sterility is compromised, increasing the risk of contamination. Even if the vial appears clean, it's crucial to discard it and use a fresh vial for each procedure to maintain patient safety.
Sterile water vials 10ml are typically made from Type I borosilicate glass, known for its chemical inertness and low leachability. Increasingly, specialized plastics designed for pharmaceutical applications are also used. The vial closure is often made of rubber stoppers coated with a protective layer to maintain sterility and prevent drug interactions.
Sterile water vials 10ml should be stored in a cool, dry place, away from direct sunlight and extreme temperatures. Avoid storing them in areas with high humidity or potential contamination. Ensure the vials remain sealed until ready for use and inspect for any signs of damage or leakage before administration.
In conclusion, sterile water vials 10ml are indispensable components of modern healthcare, offering a vital and versatile solution for a diverse range of medical applications. Their consistent quality, assured sterility, and convenient 10ml volume contribute to improved patient safety, enhanced treatment efficacy, and streamlined clinical workflows. The continued development of innovative manufacturing techniques and sustainable materials promises to further optimize their performance and environmental footprint.
Looking ahead, investing in robust supply chain infrastructure, embracing digitalization, and adhering to stringent regulatory standards will be crucial for ensuring the ongoing availability of high-quality sterile water vials 10ml. By prioritizing innovation and collaboration, the healthcare industry can leverage these essential vials to address evolving medical needs and improve global health outcomes. For reliable sourcing and a commitment to quality, visit our website: www.wkpacking.com