Small liquid medicine containers are vital components in healthcare delivery globally, ensuring safe and accurate dosage of essential medications. From over-the-counter remedies to life-saving prescriptions, these containers play a critical role in patient well-being and treatment efficacy. Understanding their design, materials, and regulatory standards is paramount for manufacturers, distributors, and healthcare professionals alike. small liquid medicine container quality directly impacts patient safety.
The demand for small liquid medicine containers is consistently high, driven by an aging population, increasing prevalence of chronic diseases, and a growing emphasis on preventative healthcare. Global health crises, like pandemics, further amplify this need, necessitating robust supply chains and innovative packaging solutions. The evolution of these containers focuses on improved child-resistance, tamper-evidence, and user-friendliness.
Effective small liquid medicine container solutions aren't just about containment; they’re about promoting adherence to treatment plans and minimizing medication errors. This necessitates a focus on clear labeling, easy-to-use dispensing mechanisms, and materials that maintain drug stability throughout their shelf life.


The landscape of pharmaceutical packaging is constantly evolving, with small liquid medicine containers at the forefront of innovation. These specialized containers are engineered to protect the integrity of liquid medications while facilitating accurate and safe dispensing for patients of all ages. small liquid medicine container solutions are therefore integral to modern healthcare.
Beyond simple containment, the design of these containers must address critical factors such as child-resistance, tamper-evidence, material compatibility, and regulatory compliance. A poorly designed container can lead to medication errors, accidental ingestion, or degradation of the drug itself, underscoring the importance of quality control and robust manufacturing processes.
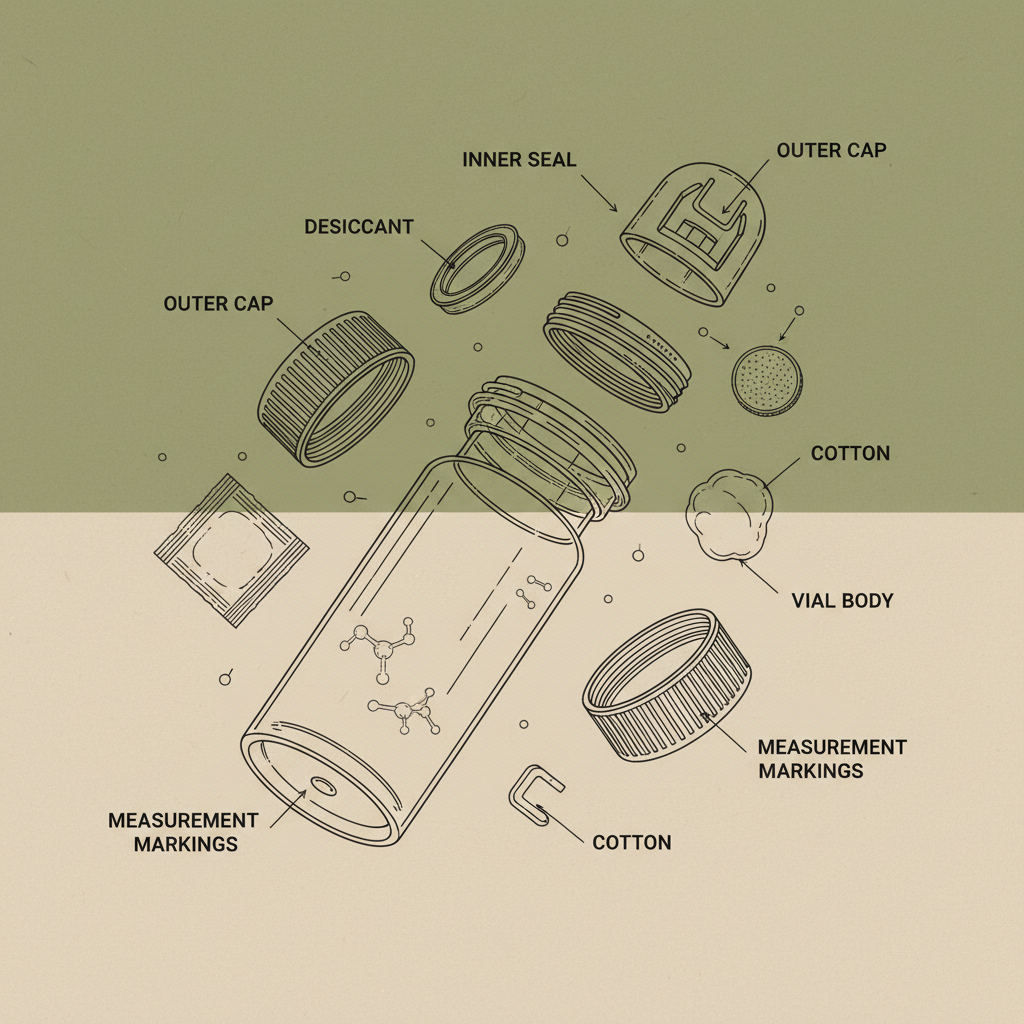
A small liquid medicine container is typically defined as a packaging system designed to hold a volume of liquid medication ranging from a few milliliters to approximately 100 milliliters. These containers are commonly constructed from materials such as polyethylene (PE), polypropylene (PP), or glass, chosen for their chemical inertness and ability to maintain drug stability. The term encompasses bottles, vials, and dropper bottles used for oral, topical, or injectable liquid formulations.
These containers are inextricably linked to modern pharmaceutical practice, enabling accurate dosing and convenient administration of medications. They are essential for ensuring patient compliance and maximizing the therapeutic benefits of prescribed treatments. The connection to humanitarian needs is also significant, as these containers facilitate the delivery of vital medicines in disaster relief and healthcare programs in developing countries.
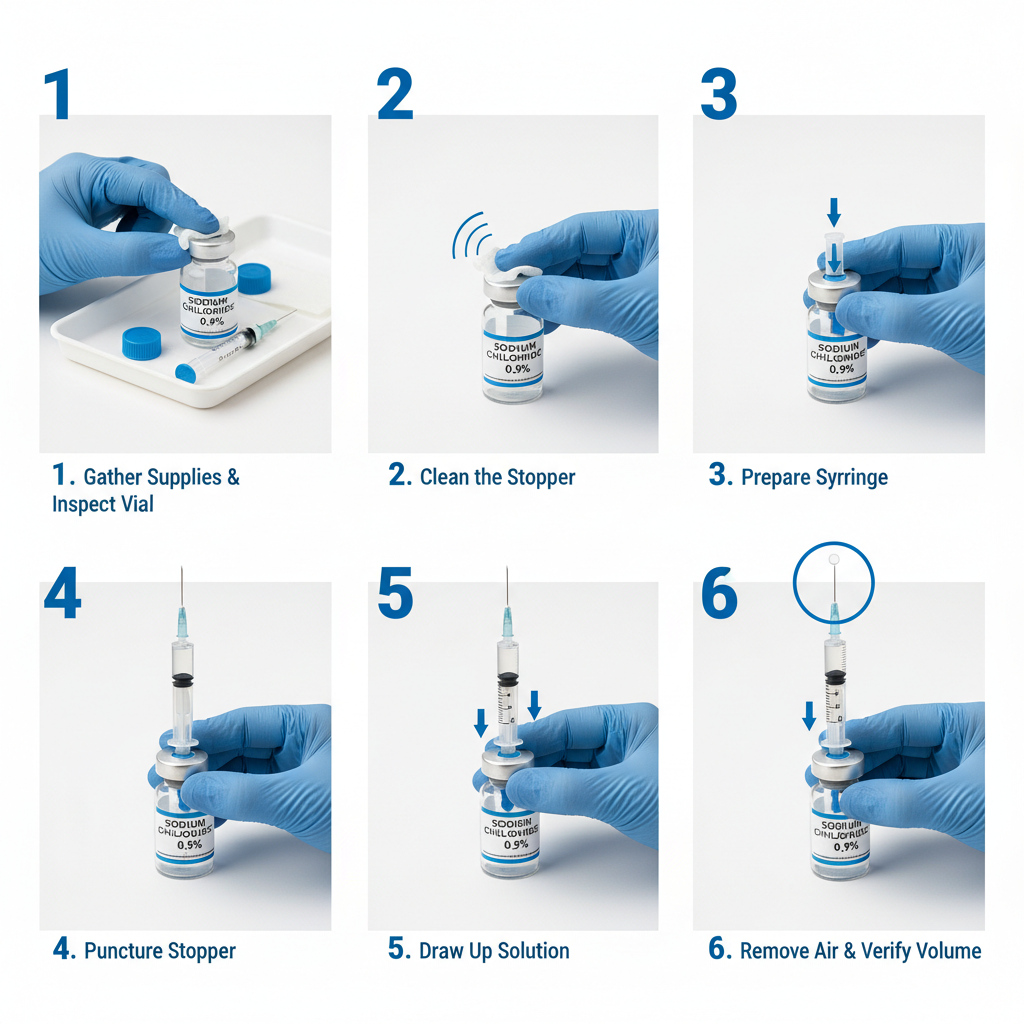
The functionality of a small liquid medicine container extends beyond simply holding the liquid; it involves precise dispensing mechanisms, often incorporating features like child-resistant closures, droppers, or calibrated measuring caps. This precision is crucial in ensuring patients receive the correct dosage, minimizing the risk of adverse effects or treatment failure.
Durability is paramount. Small liquid medicine containers must withstand the rigors of handling, transportation, and storage without compromising the integrity of the medication. This requires selecting materials with appropriate impact resistance and chemical compatibility. small liquid medicine container resilience directly affects medicine efficacy.
Child-resistance is a critical safety feature. Regulations in many countries mandate that containers for certain medications incorporate mechanisms to prevent accidental ingestion by children. These mechanisms often involve specialized closures that require a specific sequence of movements to open, making them difficult for young children to manipulate.
Material compatibility ensures that the container does not interact with the medication, causing degradation or contamination. The choice of material must consider the chemical properties of the drug, its pH, and its sensitivity to light and oxygen. Using the incorrect material can compromise the medication’s effectiveness and even create harmful byproducts.
Small liquid medicine containers find widespread application across numerous sectors, including pharmaceutical manufacturing, healthcare facilities, and retail pharmacies. They are essential for dispensing prescription medications, over-the-counter remedies, diagnostic reagents, and even nutritional supplements. The versatility of these containers makes them indispensable in a broad range of healthcare settings.
In post-disaster relief operations, lightweight and durable small liquid medicine containers are crucial for delivering essential medications to affected populations. Organizations like the World Health Organization (WHO) rely on standardized packaging to ensure efficient distribution and prevent contamination. Remote industrial zones often depend on these containers for workplace first aid and emergency medical supplies.
Developing countries benefit immensely from the availability of affordable and reliable small liquid medicine containers, allowing for wider access to essential medicines. NGOs often distribute medications in these containers as part of public health initiatives targeting infectious diseases and chronic conditions. The right container can improve medication adherence and treatment outcomes.
The advantages of using high-quality small liquid medicine containers are multifaceted. Cost-effectiveness is a key benefit, as these containers are typically manufactured from readily available and relatively inexpensive materials. Their lightweight nature also reduces shipping costs and logistical complexities.
Sustainability is increasingly important. The growing use of recyclable materials, like certain grades of polyethylene and polypropylene, minimizes environmental impact. Furthermore, the design of containers can be optimized to reduce material usage without compromising performance. From a social impact perspective, safe and reliable packaging contributes to public health and prevents medication errors, building trust in the healthcare system.
The future of small liquid medicine containers is likely to be shaped by advancements in materials science, digital technologies, and sustainable packaging practices. Biodegradable and compostable materials are gaining traction as alternatives to traditional plastics, offering a more environmentally friendly solution.
Digital integration, such as the incorporation of RFID tags or QR codes, can enhance traceability and combat counterfeiting. Smart containers equipped with sensors can monitor medication temperature and humidity, ensuring drug stability throughout the supply chain. Automation in manufacturing processes will further drive down costs and improve efficiency.
One significant challenge is maintaining child-resistance without compromising usability for elderly or disabled patients. Innovative closure designs that require minimal dexterity while remaining secure are crucial. Another challenge is the need to reduce reliance on fossil fuel-based plastics.
Developing and scaling up the production of cost-effective biodegradable alternatives is a key area of focus. Furthermore, improving recycling infrastructure and promoting consumer awareness are essential for maximizing the benefits of sustainable packaging. Collaboration between manufacturers, regulators, and healthcare providers is vital to address these challenges effectively.
Ensuring global regulatory harmonization is also critical. Different countries have varying standards for small liquid medicine container safety and performance, creating complexities for manufacturers operating in multiple markets. Standardization efforts can streamline the approval process and ensure consistent quality across borders.
| Challenge | Impact Area | Potential Solution | Implementation Difficulty (1-5) |
|---|---|---|---|
| Maintaining Child Resistance | Patient Safety | Ergonomic Closure Designs | 3 |
| Plastic Waste Generation | Environmental Sustainability | Biodegradable Material Adoption | 4 |
| Medication Degradation | Drug Efficacy | Optimized Material Compatibility | 2 |
| Counterfeit Packaging | Patient Safety & Brand Protection | Digital Tracking & Authentication | 3 |
| Regulatory Discrepancies | Manufacturing & Distribution | Harmonized Global Standards | 5 |
| Supply Chain Vulnerabilities | Healthcare Access | Diversified Sourcing & Local Production | 4 |
The most prevalent materials are Polyethylene (PE) and Polypropylene (PP) due to their chemical inertness, cost-effectiveness, and good barrier properties. Glass is also used, particularly for medications requiring a higher level of protection from oxygen or moisture. HDPE (High-Density Polyethylene) and PET (Polyethylene Terephthalate) offer specific advantages depending on the drug formulation and intended use, ensuring medication stability and patient safety.
Child-resistant packaging is critically important, mandated by regulations in many countries to prevent accidental ingestion by children. These closures require specific manipulation, such as pressing and twisting, making them difficult for young children to open. However, it’s equally important that these closures remain usable for elderly or individuals with limited dexterity, balancing safety with accessibility.
Key regulations include USP (United States Pharmacopeia) standards for container closures, as well as requirements from regulatory bodies like the FDA (Food and Drug Administration) and EMA (European Medicines Agency). These regulations cover aspects like material compatibility, leachables, extractables, and tamper-evidence. Compliance is crucial for ensuring product safety and gaining market approval.
Ensuring quality involves rigorous testing throughout the manufacturing process, including material verification, leak testing, and dimensional analysis. Good Manufacturing Practices (GMP) must be followed, and regular audits conducted to verify compliance. Utilizing reputable suppliers and implementing robust quality control systems are essential for maintaining product integrity.
Yes, there’s growing interest in biodegradable and compostable materials like PLA (Polylactic Acid) and PHA (Polyhydroxyalkanoates). Recycled plastics are also gaining traction. However, challenges remain in terms of cost, scalability, and ensuring comparable performance and barrier properties to traditional plastics. Research and development are ongoing to overcome these hurdles.
Clear and accurate labeling is crucial to prevent medication errors. This includes legible font sizes, distinct color-coding, and easily understandable instructions. Well-designed dispensing mechanisms, such as calibrated droppers or measuring caps, also help patients administer the correct dosage. Tamper-evident features reassure patients that the medication hasn’t been compromised.
In conclusion, small liquid medicine containers represent a critical component of the global healthcare infrastructure. Their design, materials, and manufacturing processes directly impact patient safety, medication efficacy, and environmental sustainability. Advancements in materials science, digital technologies, and regulatory harmonization are driving innovation in this field, paving the way for more secure, convenient, and environmentally responsible packaging solutions.
Looking ahead, a continued focus on sustainability, child-resistance, and regulatory compliance will be essential. Investing in research and development of biodegradable materials, smart packaging technologies, and robust quality control systems will unlock further improvements. Visit our website at www.wkpacking.com to explore our range of innovative small liquid medicine container solutions.