Sodium chloride 0.9 vial, commonly known as normal saline, is a ubiquitous and essential solution in healthcare and various industrial applications. Its fundamental role as an isotonic fluid makes it crucial for intravenous administration, wound irrigation, and numerous laboratory procedures. Globally, the demand for sodium chloride 0.9 vial is consistently high, driven by the expanding healthcare sector, increasing surgical procedures, and a growing awareness of hygiene and sterilization practices. sodium chloride 0.9 vial availability and quality are paramount for patient safety and effective medical care.
The significance of sodium chloride 0.9 vial extends beyond clinical settings. It serves as a vital component in pharmaceutical manufacturing, diagnostic testing, and even in certain food processing applications. The World Health Organization (WHO) emphasizes the importance of access to essential medicines, including intravenous fluids like normal saline, especially in emergency situations and resource-limited settings. Ensuring a stable supply chain and maintaining stringent quality control are key challenges in meeting global demand.
Understanding the nuances of sodium chloride 0.9 vial – its production, quality standards, and appropriate usage – is vital for healthcare professionals, researchers, and manufacturers alike. Its accessibility and reliability are integral to public health and contribute significantly to the effectiveness of medical interventions worldwide. The ongoing need for cost-effective and safe solutions fuels innovation within the manufacturing processes related to this essential fluid.
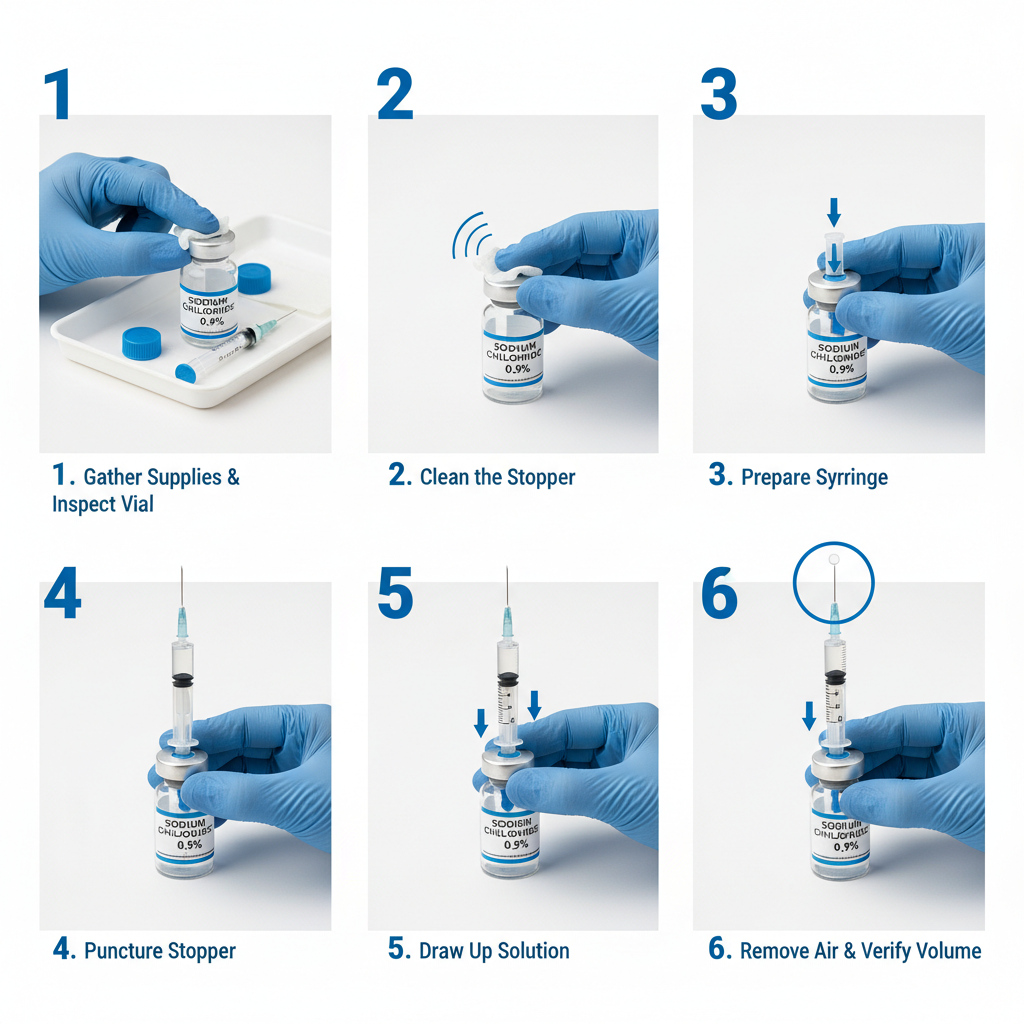

Sodium chloride 0.9 vial stands as a cornerstone of modern medical practice, providing a physiologically compatible fluid for a multitude of applications. Its simple composition belies its profound impact on patient care, ensuring hydration, medication delivery, and effective wound management. The consistent demand for this solution highlights its fundamental role in maintaining health and treating illness on a global scale. sodium chloride 0.9 vial is a crucial ingredient.
The production and distribution of sodium chloride 0.9 vial are subject to rigorous quality control standards enforced by regulatory bodies worldwide. From sourcing raw materials to final packaging, each step is carefully monitored to guarantee product purity and sterility. This dedication to quality is paramount in safeguarding patient well-being and preserving the integrity of the healthcare system.
The demand for sodium chloride 0.9 vial is inextricably linked to global healthcare trends, including an aging population, a rise in chronic diseases, and increasing access to medical care in developing countries. According to the United Nations, the global population is projected to reach 9.7 billion by 2050, further amplifying the need for essential medical supplies like normal saline. This projection underscores the importance of sustainable manufacturing and efficient distribution networks.
The COVID-19 pandemic dramatically highlighted the critical role of sodium chloride 0.9 vial in managing severely ill patients, particularly for fluid resuscitation and drug dilution. Supply chain disruptions during the pandemic exposed vulnerabilities in global manufacturing and distribution systems, prompting calls for greater diversification and resilience. The crisis underscored the necessity of maintaining strategic reserves of essential medical commodities.
International organizations, such as the World Health Organization (WHO) and UNICEF, actively support the procurement and distribution of sodium chloride 0.9 vial to regions facing humanitarian crises and healthcare shortages. Ensuring equitable access to this essential fluid is a key priority in global health initiatives aimed at reducing morbidity and mortality.
Sodium chloride 0.9 vial is an isotonic saline solution, meaning it contains 0.9% sodium chloride (NaCl) by weight, which is roughly equivalent to the salt concentration found in human blood. This isotonicity is crucial because it prevents the disruption of cellular function when administered intravenously or used for irrigation. The solution is sterile, pyrogen-free, and typically packaged in glass or plastic vials or bags for easy administration.
Its connection to modern industry and humanitarian needs lies in its versatility. It's not merely a fluid replacement but a vehicle for delivering medications, rinsing wounds to prevent infection, and maintaining electrolyte balance. In humanitarian contexts, sodium chloride 0.9 vial is often the first line of defense in treating dehydration, shock, and other life-threatening conditions following natural disasters or conflicts.
The manufacturing process involves dissolving pharmaceutical-grade sodium chloride in purified water, followed by sterilization via autoclaving or filtration. Strict adherence to pharmacopeial standards (e.g., USP, EP) is essential to ensure the product's safety and efficacy. sodium chloride 0.9 vial quality is paramount.
First, Isotonicity is a defining characteristic of sodium chloride 0.9 vial. This ensures compatibility with bodily fluids, minimizing cell damage during administration. Second, Sterility is absolutely critical; the solution must be free from any microorganisms to prevent infection. Third, Pyrogen-free status guarantees the absence of fever-inducing substances, enhancing patient safety.
Fourth, Chemical Stability allows for the secure mixing of various medications without degradation, making it ideal for IV drug delivery. Fifth, pH Neutrality (around 7.4) maintains the physiological balance when introduced into the body. Sixth, Clarity and Colorlessness indicate purity and the absence of contaminants.
In healthcare, sodium chloride 0.9 vial is used extensively for intravenous hydration, wound cleansing, drug delivery, and as a carrier fluid for blood transfusions. It plays a crucial role in emergency medicine, surgery, and intensive care units. In post-disaster relief operations, it’s often the first medical intervention, combating dehydration and shock.
Beyond medicine, sodium chloride 0.9 vial finds application in pharmaceutical manufacturing as a component in various formulations and solutions. It’s also utilized in laboratory settings for preparing solutions, calibrating instruments, and performing biological assays. In remote industrial zones, it can be essential for maintaining hygiene and treating minor injuries.
Furthermore, specific veterinary applications require the use of sodium chloride 0.9 vial for animal hydration and treatment. Its compatibility and safety make it a preferred choice for a wide range of animal health needs. sodium chloride 0.9 vial is versatile.
The primary advantage of sodium chloride 0.9 vial is its safety and biocompatibility. Its isotonicity minimizes the risk of adverse reactions, making it suitable for a broad patient population. The cost-effectiveness of production and widespread availability contribute to its long-term value, ensuring accessibility in both developed and developing countries.
From a sustainability standpoint, the manufacturing of sodium chloride 0.9 vial generally has a relatively low environmental impact compared to other complex pharmaceutical products. The long-term value extends to improved patient outcomes, reduced hospital stays, and enhanced quality of life. The feeling of security it provides to healthcare professionals is invaluable.
Emerging trends include the development of more sustainable packaging materials for sodium chloride 0.9 vial, reducing plastic waste and promoting circular economy principles. Advancements in sterilization technologies, such as low-temperature sterilization methods, aim to minimize energy consumption and environmental impact.
Digital transformation is also playing a role, with the implementation of track-and-trace systems to enhance supply chain transparency and prevent counterfeiting. Automation in manufacturing processes can improve efficiency and reduce production costs. Furthermore, research into novel delivery systems, such as microfluidic devices, could optimize drug delivery using sodium chloride 0.9 vial as a carrier.
Green chemistry principles are being applied to minimize waste and energy consumption during production. The focus on sustainability extends to responsible sourcing of raw materials and minimizing the carbon footprint associated with transportation and distribution.
A major challenge is maintaining the stability of the supply chain, particularly in regions prone to political instability or natural disasters. Counterfeiting and substandard products pose a threat to patient safety and erode public trust. Ensuring consistent quality control across diverse manufacturing facilities is also a significant hurdle.
Solutions include diversifying sourcing options, implementing robust quality assurance systems, and leveraging blockchain technology for supply chain tracking. Strengthening regulatory oversight and international collaboration can combat counterfeiting. Investing in advanced manufacturing technologies and training programs can enhance production efficiency and quality. sodium chloride 0.9 vial safety is key.
The following table summarizes common challenges and potential solutions related to sodium chloride 0.9 vial.
| Challenge | Impact Area | Severity (1-10) | Potential Solution |
|---|---|---|---|
| Supply Chain Disruptions | Availability & Cost | 8 | Diversified Sourcing & Strategic Reserves |
| Counterfeit Products | Patient Safety & Trust | 9 | Blockchain Tracking & Regulatory Enforcement |
| Quality Control Variability | Efficacy & Compliance | 7 | Standardized Protocols & Audits |
| Packaging Waste | Environmental Impact | 6 | Sustainable Materials & Recycling Programs |
| Sterilization Costs | Production Costs | 5 | Low-Temperature Sterilization Technologies |
| Regulatory Complexity | Compliance & Market Access | 7 | Harmonized Standards & Global Collaboration |
Generally, an unopened sodium chloride 0.9 vial has a shelf life of several years, typically 2-5 years, depending on the manufacturer and packaging. However, it's crucial to always check the expiration date printed on the vial. The integrity of the packaging is also vital; any damage should render the vial unusable. Proper storage, away from direct sunlight and extreme temperatures, is essential to maintain stability.
Yes, sodium chloride 0.9 vial is commonly used to dilute medications for intravenous administration. Its isotonicity makes it a compatible diluent for many drugs, minimizing the risk of hemolysis or other adverse reactions. However, compatibility should always be verified with a pharmacist or drug information resource before mixing any medication. It's important to follow established protocols for drug dilution to ensure accurate dosage.
Using contaminated sodium chloride 0.9 vial can lead to serious infections, fever, sepsis, and potentially life-threatening complications. Contamination can occur during manufacturing, packaging, or handling. Strict adherence to sterile techniques is paramount when administering the solution. Signs of contamination, such as cloudiness or particulate matter, should immediately disqualify the vial for use.
Sodium chloride 0.9 vial is manufactured under stringent sterile conditions. The process typically involves dissolving pharmaceutical-grade sodium chloride in purified water, followed by sterilization using autoclaving (high-pressure steam) or filtration through sterile filters with pore sizes small enough to remove bacteria and other microorganisms. Regular quality control testing is conducted to verify sterility and the absence of pyrogens.
Alternatives include lactated Ringer's solution, which contains electrolytes in addition to sodium chloride, and dextrose solutions. The choice of IV fluid depends on the patient's specific needs and medical condition. Sodium chloride 0.9 vial is often preferred for initial fluid resuscitation and when electrolyte balance is not a primary concern.
Sodium chloride 0.9 vial should be stored in a cool, dry place, protected from direct sunlight and extreme temperatures. It's important to keep the vials in their original packaging until use. Avoid freezing the solution, as this can compromise its stability. Check the manufacturer's instructions for specific storage recommendations.
Sodium chloride 0.9 vial remains an indispensable component of modern healthcare and a vital resource in various industrial applications. Its inherent safety, versatility, and cost-effectiveness solidify its position as a cornerstone of medical practice. Understanding its properties, applications, and potential challenges is critical for ensuring optimal patient care and maintaining a resilient supply chain.
Looking ahead, innovation in sustainable packaging, sterilization techniques, and digital tracking will further enhance the value of sodium chloride 0.9 vial. Continuous investment in quality control and adherence to stringent regulatory standards are essential to safeguarding patient safety and fostering public trust. Visit our website at www.wkpacking.com to learn more about our products and services.

