10 ml sterile water vials are fundamental components in a vast array of scientific, medical, and industrial applications. Their critical role stems from the necessity of having a readily available, contamination-free source of water for injection, dilution, rinsing, and various analytical procedures. Understanding their manufacturing processes, quality control measures, and diverse applications is paramount for ensuring the integrity and reliability of countless processes globally.
The global demand for 10 ml sterile water vials is steadily increasing, driven by the expanding pharmaceutical industry, advancements in biotechnology, and the growing emphasis on stringent hygiene standards across healthcare sectors. According to the World Health Organization, access to safe water for injection is crucial for preventing healthcare-associated infections, a significant global health challenge. These vials play a critical role in meeting that demand.
Furthermore, the availability of consistently high-quality 10 ml sterile water vials is crucial not only for established healthcare systems but also for resource-limited settings, where access to essential medicines and safe injection practices can be severely compromised. This underscores the importance of reliable supply chains and adherence to international quality standards like those set by ISO.


The use of 10 ml sterile water vials is deeply embedded within modern healthcare and laboratory practices. They represent a cornerstone of maintaining aseptic conditions, vital for accurate testing, drug preparation, and safe patient care. The demand stems from the increasing emphasis on patient safety and the need for reliable, contamination-free consumables.
These vials are not merely containers of water; they are carefully manufactured and rigorously tested products, adhering to strict pharmaceutical standards. Their consistent quality directly impacts the validity of research findings, the efficacy of medications, and the safety of medical procedures performed worldwide.
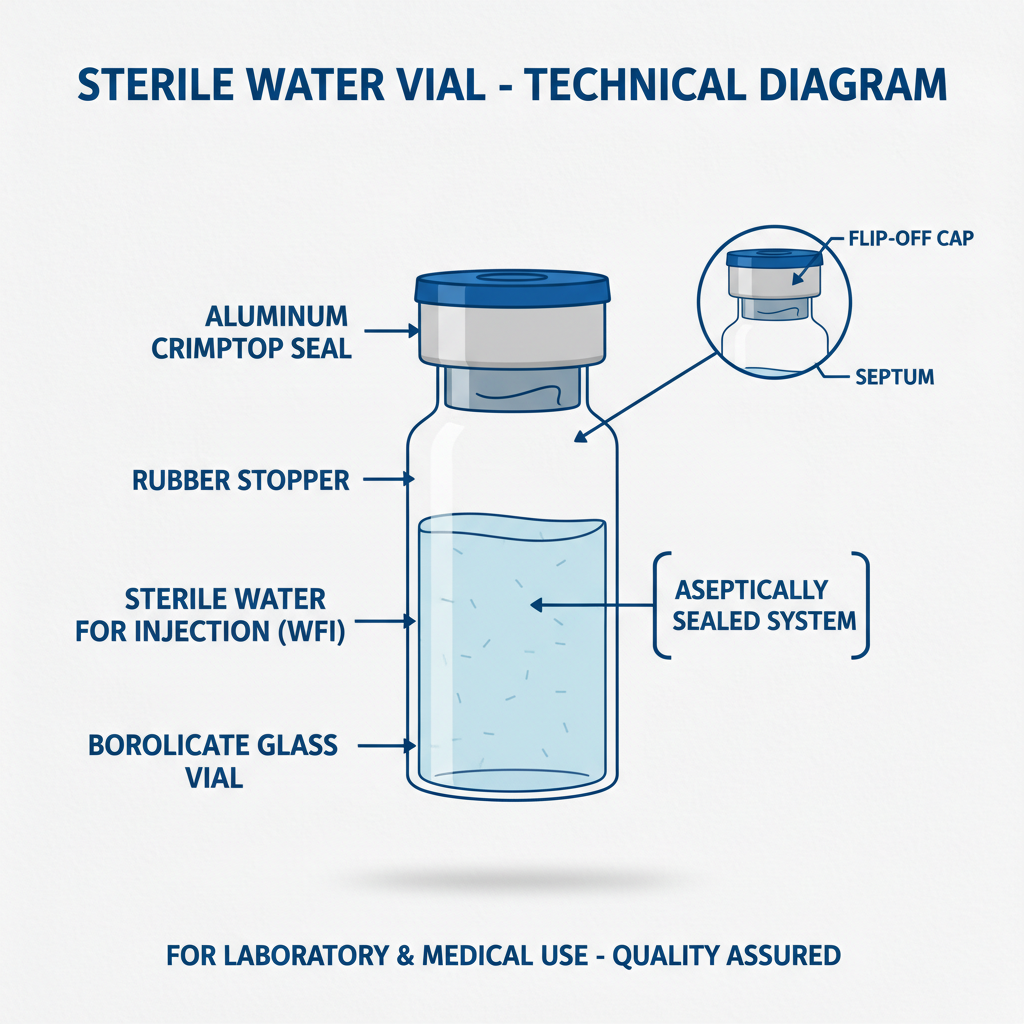
10 ml sterile water vials are typically made of pharmaceutical-grade glass or plastic, designed to hold 10 milliliters of Water for Injection (WFI). This water undergoes a meticulous purification process, including distillation, deionization, and often, filtration, to remove all microorganisms, pyrogens, and particulate matter. The vials themselves are sterilized through methods like autoclaving or dry heat sterilization.
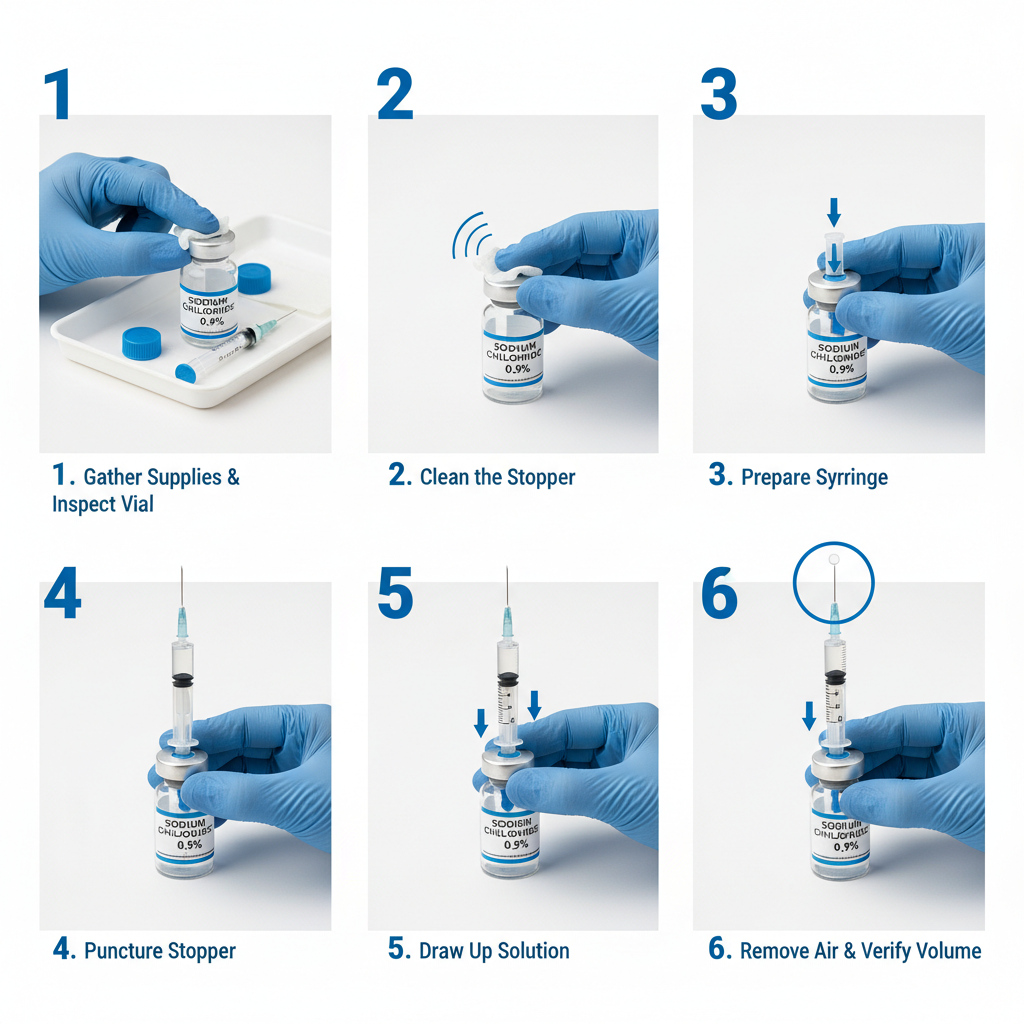
These vials are fundamentally important for applications where purity and the absence of microbial contamination are non-negotiable. They are critical in compounding pharmacies for reconstituting injectable medications, in clinical laboratories for preparing samples for analysis, and in research settings for cell culture and other sensitive experiments.
The term "sterile" signifies the complete absence of viable microorganisms, a requirement dictated by pharmacopoeial standards like those established by the United States Pharmacopeia (USP) and the European Pharmacopoeia (EP). Consistent adherence to these standards is essential to ensure the safety and efficacy of the products utilizing these vials.
The integrity of 10 ml sterile water vials hinges on several key characteristics. Firstly, 10 ml sterile water vials must exhibit exceptional chemical inertness, ensuring no leaching of substances from the vial material into the water, which could compromise its purity. Secondly, they must provide an effective barrier against microbial ingress, maintaining sterility over their shelf life.
Furthermore, the physical properties of the vials, such as glass type or plastic composition, impact their resistance to breakage and their compatibility with various storage and handling conditions. Proper sealing mechanisms, typically employing rubber stoppers and aluminum crimp seals, are crucial for maintaining both sterility and preventing evaporation. The choice of stopper and seal material also factors into the overall chemical compatibility.
Finally, the manufacturing process itself is rigorously controlled, with extensive quality control testing at each stage to ensure compliance with regulatory standards. This includes testing for particulate matter, endotoxins, and sterility to guarantee product safety and efficacy.
The applications of 10 ml sterile water vials are remarkably diverse. Within the pharmaceutical industry, they’re indispensable for diluting medications for intravenous administration, reconstituting lyophilized drugs, and preparing sterile solutions for research and development. 10 ml sterile water vials are routinely used in compounding pharmacies, where individualized medications are prepared based on specific patient needs.
In clinical diagnostics, these vials serve as a crucial component in preparing samples for various analyses, including hematology, clinical chemistry, and microbiology. Maintaining the sterility of these samples is critical for accurate results and reliable diagnoses. The biotechnology sector also heavily relies on these vials for cell culture, media preparation, and other sensitive experiments.
The demand for 10 ml sterile water vials is globally distributed, with significant consumption in regions with well-developed healthcare systems, such as North America, Europe, and Japan. However, rapidly growing pharmaceutical markets in emerging economies, including India, China, and Brazil, are driving increased demand for these vials.
In post-disaster relief operations, 10 ml sterile water vials are critical for providing safe hydration and administering essential medications in challenging field conditions. Organizations like the World Health Organization (WHO) and Doctors Without Borders rely heavily on readily available sterile water sources for emergency medical care.
Utilizing high-quality 10 ml sterile water vials offers numerous advantages. Cost-effectively providing a consistently pure and sterile water source minimizes the risk of contamination, contributing to patient safety and treatment efficacy. The reliability of these vials reduces the potential for treatment failures and associated healthcare costs.
From a social impact perspective, readily available sterile water vials support equitable access to essential healthcare services, particularly in resource-limited settings. The emotional reassurance of safe and reliable medications fosters trust in the healthcare system and promotes better patient outcomes.
Long-term, investing in robust quality control processes and sustainable manufacturing practices for these vials builds brand reputation and strengthens supply chain resilience, enhancing trust among healthcare providers and ultimately benefiting patients globally.
Innovation in 10 ml sterile water vial technology is focused on several key areas. Development of more sustainable materials, such as bio-based plastics, aims to reduce the environmental impact of vial manufacturing and disposal. Advanced sterilization techniques, like vaporized hydrogen peroxide (VHP) sterilization, are being explored to enhance sterility assurance.
Digital technologies, including blockchain, are being implemented to improve supply chain traceability and combat counterfeiting, ensuring the authenticity and quality of these vials. Automation in manufacturing processes is increasing efficiency and reducing production costs.
Furthermore, integrating smart sensors into vial packaging could provide real-time monitoring of temperature and humidity, ensuring the integrity of the sterile water throughout its storage and transportation.
| Innovation Area | Current Status | Potential Impact | Implementation Challenges |
|---|---|---|---|
| Sustainable Materials | Early Stage R&D - Bio-based plastics | Reduced environmental footprint | Cost & Performance |
| Advanced Sterilization | VHP gaining traction | Enhanced sterility assurance | Equipment Costs |
| Supply Chain Traceability | Blockchain pilot programs | Combating counterfeiting | Data integration |
| Automated Manufacturing | Increasingly adopted | Improved efficiency & reduced costs | Initial investment |
| Smart Packaging | Concept Stage – Sensor integration | Real-time condition monitoring | Sensor cost & data security |
| Enhanced Quality Control | AI-driven inspection systems | Improved defect detection | Algorithm development |
The shelf life of a 10 ml sterile water vial typically ranges from 2 to 5 years, provided it is stored correctly – usually in a cool, dry place away from direct sunlight. However, the specific expiration date is determined by the manufacturer and printed on the vial label. Always check the expiration date before use, as sterility cannot be guaranteed beyond this date. Proper storage prevents degradation of the vial material and maintains the integrity of the sterile water.
10 ml sterile water vials are most commonly made from Type I borosilicate glass, known for its exceptional chemical inertness and low leachability. Increasingly, certain applications are utilizing cyclic olefin copolymer (COC) or polypropylene plastics, particularly for single-use applications or where glass breakage is a concern. The stopper used to seal the vial is typically made of bromobutyl rubber, selected for its ability to maintain a tight seal while minimizing interactions with the water.
Sterility is ensured through a rigorous process that includes multiple stages. Initially, the Water for Injection (WFI) is thoroughly purified and filtered. Then, vials are sterilized using methods such as autoclaving (steam sterilization under pressure) or dry heat sterilization. Post-sterilization, vials undergo stringent quality control testing, including sterility testing, to confirm the absence of viable microorganisms. This testing adheres to pharmacopoeial standards like USP and EP.
Quality control tests include visual inspection for defects, particulate matter testing using light obscuration or microscopy, endotoxin testing (using the Limulus Amebocyte Lysate – LAL – test), pH testing, conductivity testing, and sterility testing. Leak testing is also conducted to ensure the integrity of the seal. These tests verify that the vials meet stringent pharmacopoeial standards and are safe for their intended use.
Yes, several seal types are available. The most common is a bromobutyl rubber stopper secured with an aluminum crimp seal. Fluoropolymer-coated stoppers are used for sensitive medications to minimize interactions. Flip-off seals offer easy access for syringe insertion, and press-on seals provide a tamper-evident closure. The choice of seal depends on the specific application and the characteristics of the drug or solution being contained.
The manufacturing of 10 ml sterile water vials is heavily regulated by international standards such as those set forth by the United States Pharmacopeia (USP), the European Pharmacopoeia (EP), and various national regulatory agencies like the FDA in the US. These standards dictate requirements for materials, manufacturing processes, sterilization methods, quality control testing, and labeling, ensuring product safety and efficacy.
In conclusion, 10 ml sterile water vials represent a crucial, yet often understated, component of modern healthcare and scientific research. Their consistent quality, assured sterility, and diverse applications underpin a wide range of critical procedures, from drug administration to laboratory analysis. Understanding the nuances of their manufacturing, quality control, and evolving technologies is paramount for maintaining the integrity of these vital supplies.
Looking ahead, continued innovation in sustainable materials, advanced sterilization techniques, and enhanced supply chain traceability will further solidify the role of 10 ml sterile water vials in safeguarding public health and advancing scientific discovery. We invite you to explore our comprehensive range of high-quality vials and related products at www.wkpacking.com.