Distilled water vials represent a critical component in numerous scientific, medical, and industrial applications. Their role extends beyond simple containment, impacting the reliability and accuracy of processes ranging from pharmaceutical manufacturing to delicate laboratory experiments. The demand for high-quality distilled water vials is steadily increasing globally, driven by advancements in these sectors and heightened quality control standards. Understanding the nuances of these vials – from their material composition to sterilization protocols – is paramount for professionals and researchers alike.
Globally, the market for distilled water vials is heavily influenced by the expansion of healthcare infrastructure, particularly in emerging economies. The pharmaceutical industry remains a dominant consumer, requiring sterile, inert vials for injectable drugs and vaccine storage. Furthermore, the growing emphasis on environmental monitoring and water quality analysis fuels the demand for vials used in sample collection and transportation. This demand is further propelled by stringent regulations set by organizations like the ISO and regulatory bodies ensuring purity and safety.
These vials aren’t merely containers; they’re integral to maintaining the integrity of sensitive substances. From ensuring the sterility of injectable medications to preserving the accuracy of scientific samples, distilled water vials play a crucial, often unseen, role in countless processes that impact our daily lives. The precision and reliability they offer are non-negotiable in fields where even the smallest contamination can have significant consequences.

The use of distilled water vials is widespread across sectors demanding high levels of purity and sterility. From storing samples for critical medical analysis to acting as containers for reactive chemicals, these vials serve as an essential barrier against contamination. The selection of the appropriate distilled water vial – considering factors like material, closure type, and sterilization method – directly impacts the reliability of subsequent processes.
The global market for these vials is driven by the relentless pursuit of accuracy and safety in research, healthcare, and industrial manufacturing. Improvements in materials science and manufacturing techniques are continuously refining the performance and accessibility of distilled water vials, further solidifying their indispensable role in modern science.
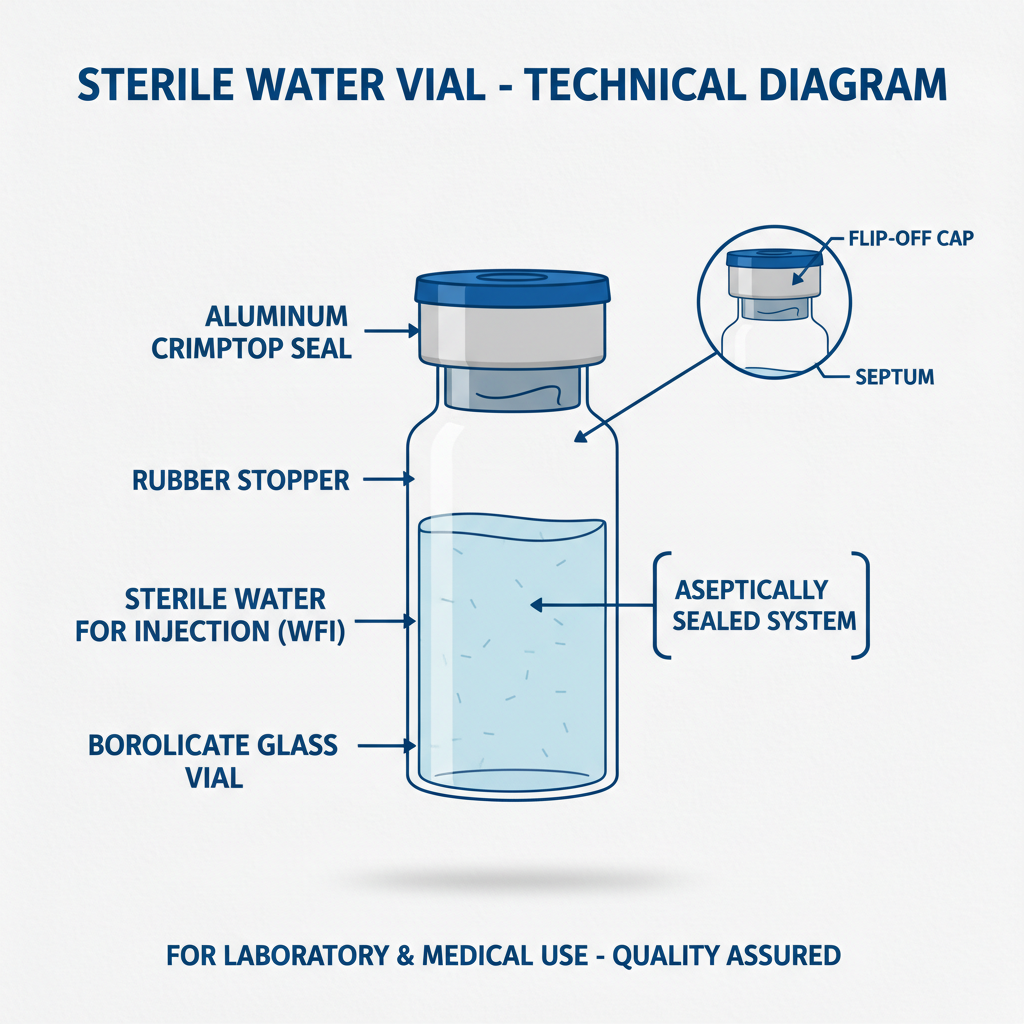
A distilled water vial is a specialized container designed to hold liquids, typically distilled water or other sterile solutions, while maintaining their purity and preventing contamination. They are commonly constructed from borosilicate glass, known for its chemical inertness and thermal resistance, though plastic options like polypropylene are also available for specific applications. Their primary function is to securely store and transport fluids without introducing impurities.
These vials aren’t simply vessels; they are meticulously engineered components supporting modern industry and humanitarian needs. In medical settings, they guarantee the safety of injectable drugs, while in environmental science, they provide a means to accurately collect and analyze water samples. The demand for these vials underscores the fundamental need for reliable containment in a world increasingly focused on precision and health.
The defining characteristic of a distilled water vial is its ability to maintain the integrity of its contents. This often requires specialized closures, like Teflon-lined caps, to prevent leaching and ensure a tight seal. Sterilization methods, such as autoclaving or gamma irradiation, are routinely applied to eliminate any microbial presence, ensuring the liquid remains free from contamination.
Several key factors contribute to the quality and suitability of a distilled water vial for a given application. Firstly, material composition is crucial. Borosilicate glass offers excellent chemical resistance and is commonly preferred for sensitive solutions. Secondly, sterility assurance is paramount, requiring validated sterilization processes and stringent quality control measures.
Closure integrity is another vital aspect, preventing leaks and contamination. Teflon-lined caps are often favored for their chemical inertness and secure seal. Finally, dimensional accuracy is important for automated filling and dispensing systems, ensuring precise volumes are consistently delivered. A poorly manufactured vial can lead to significant errors in scientific experiments or pharmaceutical production.
Additionally, manufacturing consistency is critical to ensure uniformity in performance. Variations in glass thickness or closure fit can compromise sterility and affect the reliability of results. The ability of a vial to withstand temperature fluctuations without cracking or leaching is also a crucial factor, particularly in applications involving freezing or heating.
Distilled water vials find widespread application across a diverse range of industries. In the pharmaceutical industry, they are indispensable for storing and distributing injectable drugs, vaccines, and sterile solutions. Their inert nature prevents interaction with the medication, ensuring efficacy and safety. In clinical laboratories, they’re used for collecting and analyzing blood, urine, and other bodily fluids.
The environmental monitoring sector relies heavily on distilled water vials for collecting water samples for analysis of pollutants and contaminants. In research and development, particularly in fields like chemistry and biology, they are used to store reagents, standards, and experimental solutions. Even in the cosmetics industry, sterile vials are used to preserve the purity of sensitive formulations.
The benefits of utilizing high-quality distilled water vials extend beyond simple containment. From a cost perspective, bulk purchasing and optimized designs can contribute to significant savings. In terms of sustainability, glass vials are recyclable, reducing environmental impact. Socially, using sterile vials protects public health, particularly in healthcare settings.
Moreover, the reliability of these vials is paramount. A compromised vial can invalidate research results or compromise the safety of a medication. The emotional peace of mind knowing samples are securely contained and protected is an often-overlooked advantage. Investing in quality distilled water vials builds trust in the integrity of the processes they support.
The future of distilled water vial technology is leaning towards sustainable materials and innovative designs. Biodegradable plastic alternatives are being explored to reduce environmental impact. Automated filling and capping systems, integrated with vial tracking technologies, are enhancing efficiency and minimizing human error.
Digital integration, such as RFID tagging for vial identification and temperature monitoring, will likely become increasingly common. Furthermore, advancements in glass manufacturing are enabling the production of thinner, lighter vials without compromising strength or sterility. These innovations reflect a growing commitment to sustainability and precision in all aspects of vial production and usage.
One major challenge in distilled water vial production is maintaining consistently high sterility levels. This requires rigorous quality control processes and validated sterilization techniques. Another hurdle is reducing manufacturing costs while preserving vial integrity. Finding the right balance between affordability and performance is crucial.
Solutions include investing in advanced automation technologies, optimizing manufacturing processes, and exploring alternative materials. Implementing robust supply chain management systems can also help ensure a consistent supply of high-quality materials. Furthermore, collaboration between vial manufacturers, researchers, and end-users can drive innovation and address emerging challenges.
Another challenge lies in adapting to evolving regulatory requirements. Vial manufacturers must remain vigilant in complying with changing standards for sterility, material composition, and labeling. Proactive engagement with regulatory bodies and continuous improvement of quality control systems are essential for long-term success.
| Challenge | Impact on Vial Quality | Proposed Solution | Implementation Cost (1-10) |
|---|---|---|---|
| Maintaining Sterility | Compromised sample/product integrity | Enhanced quality control, validated sterilization cycles | 7 |
| High Manufacturing Costs | Limited accessibility for smaller labs | Automation, process optimization | 8 |
| Material Variability | Inconsistent vial strength/chemical resistance | Strict supplier qualification, material testing | 6 |
| Regulatory Compliance | Production delays, potential recalls | Proactive monitoring, regular audits | 9 |
| Closure Integrity Issues | Leakage, contamination risk | Improved cap design, rigorous leak testing | 5 |
| Scaling Production | Difficulty meeting increased demand | Investment in additional manufacturing capacity | 7 |
Distilled water vials are most commonly made from borosilicate glass due to its exceptional chemical inertness, thermal resistance, and ability to maintain purity. This type of glass doesn’t leach substances into the contained liquid, critical for sensitive applications. While borosilicate glass is dominant, certain applications utilize polypropylene or other compatible plastics, particularly where breakage is a concern, though these materials require careful selection to ensure they do not interact with the stored liquid. Proper material selection ensures the integrity of samples and solutions.
Distilled water vials undergo stringent sterilization processes to eliminate any microbial contamination. Common methods include autoclaving (using high-pressure steam), gamma irradiation, and ethylene oxide sterilization. Autoclaving is often the preferred method for glass vials, while gamma irradiation is suitable for both glass and plastic. The chosen method must be validated to ensure complete sterilization and compatibility with the vial material. Each method has specific parameters that must be carefully controlled to maintain vial integrity and achieve the necessary sterility assurance level.
While both appear similar, a distilled water vial is specifically designed and manufactured to ensure extremely high purity. This involves using premium materials, such as high-quality borosilicate glass, and implementing rigorous cleaning and sterilization protocols. Regular glass vials may not meet the same stringent standards for chemical inertness and sterility, making them unsuitable for sensitive applications requiring pristine conditions. Distilled water vials also often feature specialized closures, like Teflon-lined caps, to prevent contamination, a feature not typically found in standard vials.
While plastic distilled water vials (typically polypropylene) offer advantages like reduced breakage risk and lower cost, they aren't suitable for all applications. Certain chemicals can leach from the plastic into the stored liquid, compromising its purity. Therefore, plastic vials are best suited for solutions compatible with polypropylene. Glass vials generally offer better chemical inertness and are preferred for highly sensitive or reactive substances. It’s crucial to verify the chemical compatibility of the plastic material with the intended contents before use.
To ensure vial integrity during shipping, verify the packaging is intact and undamaged upon arrival. Look for signs of crushing or puncture. Inspect the vials themselves for cracks or chips. If possible, request vials be shipped in protective packaging like foam inserts or bubble wrap. If vials are essential for time-sensitive experiments, consider utilizing shipping methods with real-time tracking and temperature monitoring to further safeguard their condition. Documentation of the shipping process can also be helpful for any potential claims.
Distilled water vials come in a range of sizes to accommodate different volumes. Common sizes include 2ml, 5ml, 10ml, 20ml, and 30ml. The selection depends on the specific application and the amount of liquid being stored or transported. Smaller vials are often preferred for individual samples or aliquots, while larger vials are used for bulk storage. Specialized sizes may also be available upon request for unique requirements, but standard sizes are usually readily available and cost-effective.
Distilled water vials, though seemingly simple, are critical components underpinning a wide array of scientific, medical, and industrial processes. Their ability to maintain purity, prevent contamination, and ensure sample integrity is paramount for accurate results and safe products. From pharmaceutical manufacturing to environmental monitoring, these vials play an indispensable role in advancing knowledge and protecting public health. Understanding their properties, applications, and future trends is essential for professionals in these fields.
As technology advances and sustainability becomes increasingly important, the future of distilled water vial development will likely focus on innovative materials, optimized designs, and enhanced quality control. Investing in high-quality vials and staying abreast of the latest advancements will not only improve operational efficiency but also contribute to a safer and more sustainable future. For all your distilled water vial needs, visit our website: www.wkpacking.com.