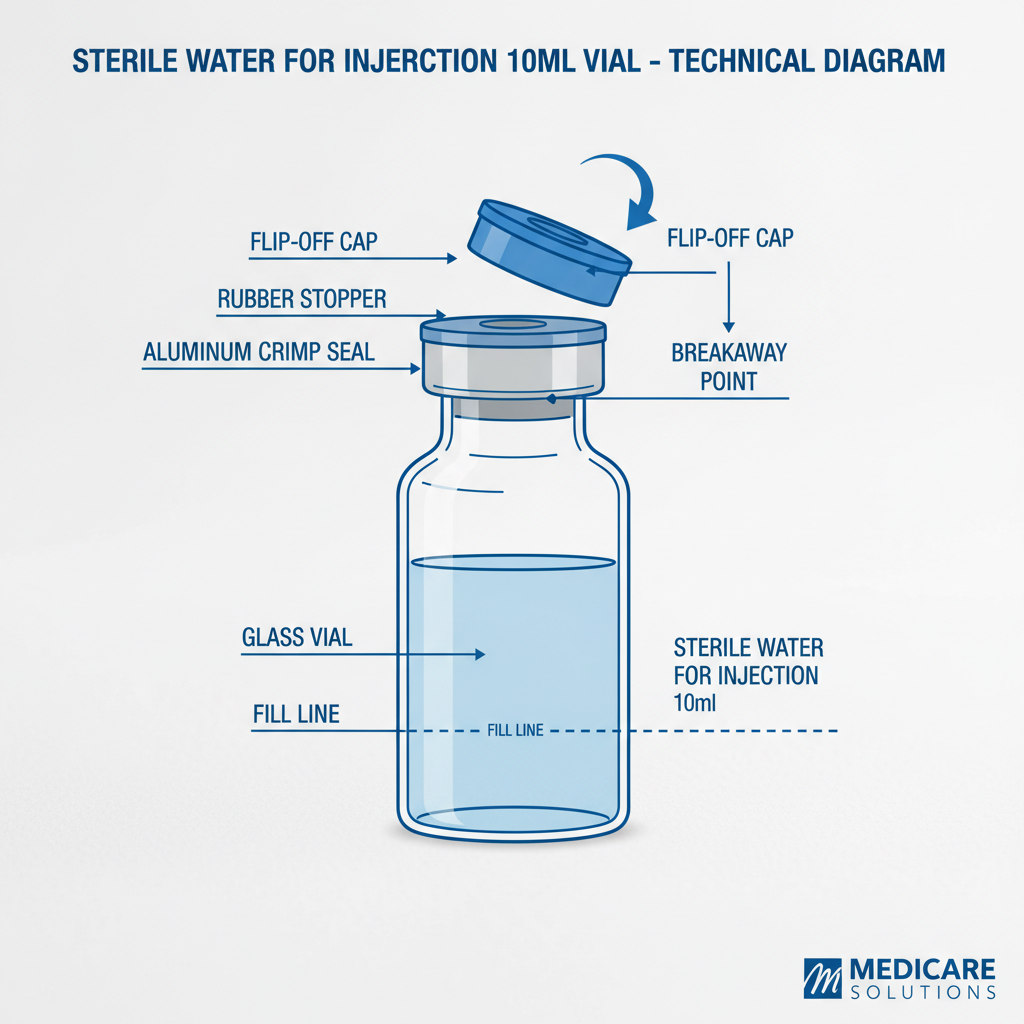
You know, things are moving fast these days. Everybody's talking about sustainability, lightweighting, and, of course, traceability. Traceability… that's a big one. Clients want to know where every single gram of plastic in a plastic vaccine bottle comes from. Honestly, it's a headache, but it's the way things are going. The demand for higher quality, more reliable packaging, especially for something as critical as vaccines, is just skyrocketing. It’s not just about protecting the product anymore; it's about building trust. And trust, well, that's everything.
It’s funny, you spend all this time designing something perfect on a computer, then you get out to a factory in, say, India, and suddenly all those elegant curves and tolerances… they just don’t translate. Have you noticed that? They look great in renders, but they’re a nightmare to mass-produce. I encountered this at a factory in Guangdong last time. The mold they had was… let’s just say it wasn’t up to spec. We had to redesign part of the bottle just to make it workable.
The plastic itself… that’s where things get interesting. We mostly use polypropylene – PP – for plastic vaccine bottles. It's relatively inexpensive, chemically resistant (crucial, obviously), and doesn't shatter easily. But the grade of PP matters. Cheap stuff smells… well, cheap. A slightly oily smell. And it feels brittle. The good stuff, though… it’s almost waxy to the touch. A little bit flexible, almost rubbery. It’s the small details, right? That’s what separates a decent bottle from a good one.
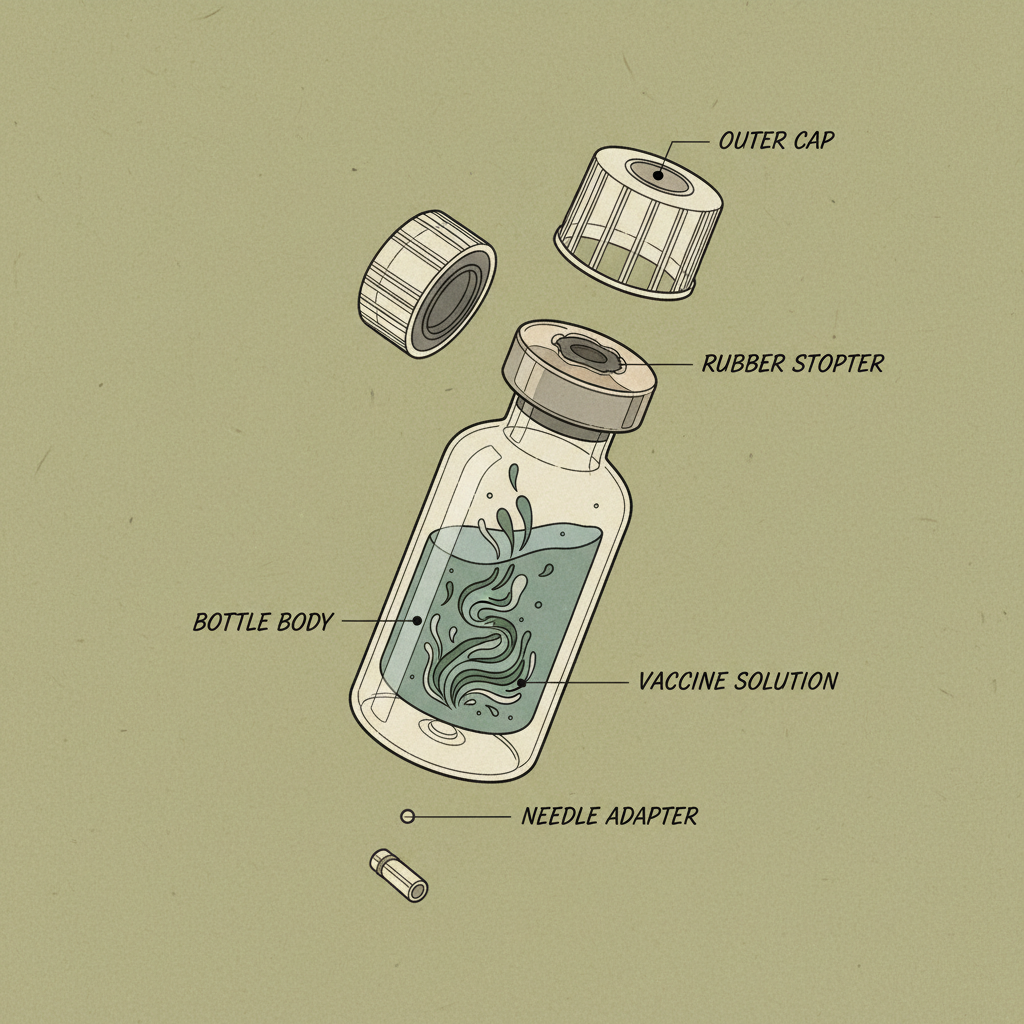
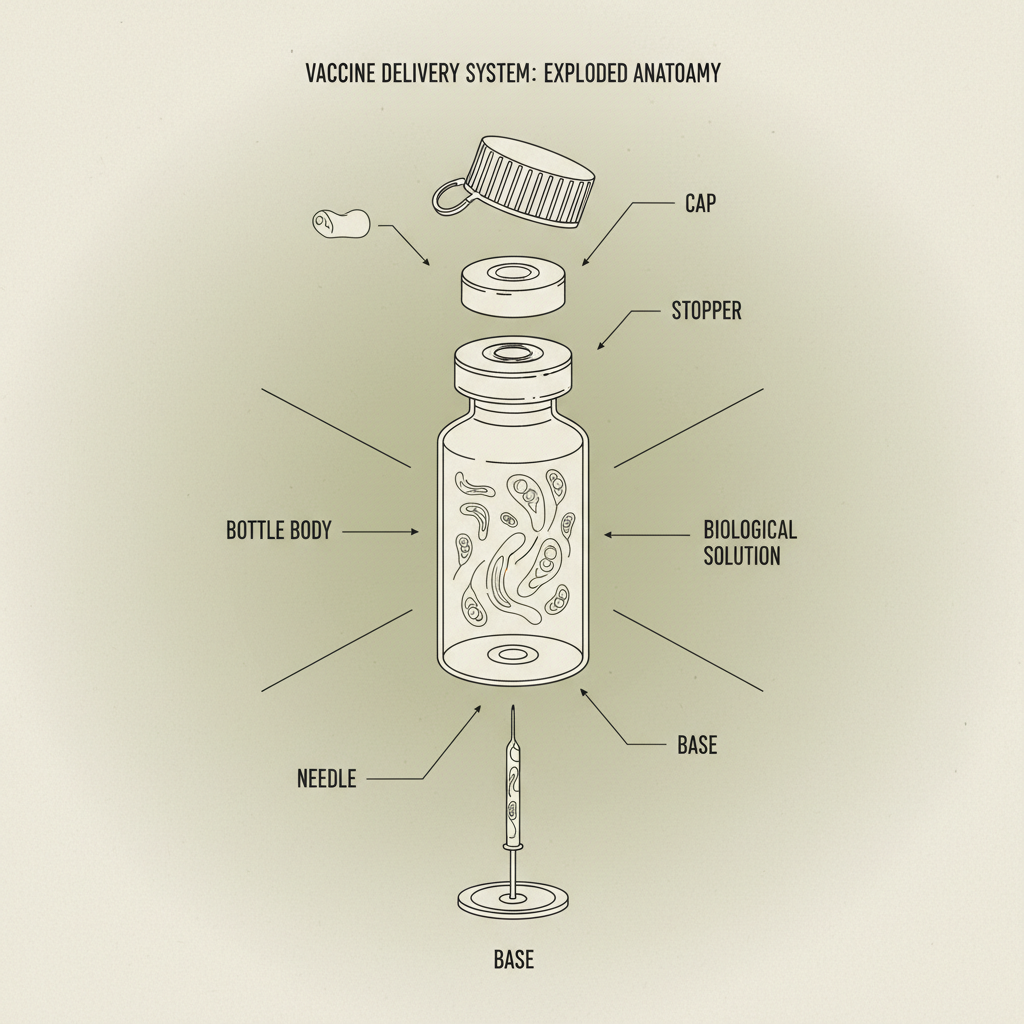
Honestly, the biggest trend right now is moving away from purely cost-driven solutions. Used to be, it was all about the cheapest possible material. Now, it’s about balance. A plastic vaccine bottle needs to be affordable, yes, but it also needs to perform. There’s been a huge push for improved barrier properties – keeping oxygen out, maintaining sterility. Strangely, a lot of manufacturers don’t fully understand the impact of different closures on the overall package integrity. You can have the best bottle in the world, but if the cap leaks, it’s all for nothing.
We’re also seeing a lot of demand for bottles that are compatible with automated filling lines. Precision is key. The dimensions have to be exactly right, or the whole system jams. It’s a little thing, but it can save manufacturers a fortune in downtime.
One thing I’ve learned over the years is to always consider the shoulder of the bottle. That’s the part that connects the body to the neck. If it’s too thin, it’ll buckle under pressure during filling or transportation. And if it’s too thick… well, it adds weight and cost. It’s a delicate balance. Another common mistake is designing a neck finish that’s too complex. Simple is almost always better. Fewer threads mean fewer points of failure. It sounds basic, but you'd be surprised...
People get obsessed with aesthetics. “It needs to look sleek!” they say. But I’m thinking, "Does it actually work?” You can’t compromise functionality for looks. It just doesn't make sense. We had a client who wanted a square bottle. A square bottle. Looked great in the mock-up, but it was a disaster in reality. The corners were stress points, and the bottles kept cracking.
And don’t even get me started on tamper-evident seals. They’re essential, of course, but they have to be easy for end-users (doctors, nurses, pharmacists) to remove. Otherwise, you end up with people stabbing at them with knives, potentially damaging the contents. It’s a usability thing, and it’s often overlooked.
Okay, so we talked about PP. But it’s not just about PP. There’s also HDPE (High-Density Polyethylene) which is tougher, but less chemically resistant. We occasionally use PET (Polyethylene Terephthalate), mainly for smaller bottles. PET has excellent clarity, which is important for visual inspection, but it's more expensive. Anyway, I think understanding the nuances of each material is critical.
And it’s not just the base polymer. It’s the additives. UV stabilizers to prevent degradation from sunlight, antioxidants to prevent oxidation, slip agents to make the bottles easier to handle… all that stuff adds up. To be honest, figuring out the right formulation can be a real headache. It’s a lot of trial and error, a lot of testing.
I saw one company trying to use recycled plastic for plastic vaccine bottles. Good intention, but the material was so inconsistent in quality that the bottles were failing quality control left and right. You can’t just swap in recycled plastic without carefully evaluating its properties. You've gotta do your homework.
Forget the lab tests, honestly. Those are important for baseline data, sure. But the real test is dropping the bottle from a reasonable height onto a concrete floor. I mean, realistically, these things are going to get banged around during shipping and handling. We also do leak tests, obviously. But we don’t just sit them on a shelf. We vibrate them, we shake them, we put them through a simulated transport environment.
One of the best tests we do is the "fill-finish-inspection" simulation. We actually fill the bottles with saline solution, cap them, label them, and then inspect them under the same conditions as the end-users would. It reveals a surprising amount of issues that you’d never catch in a lab.
You know, you design these bottles with a specific use in mind, but people always find new ways to use them. I once saw a nurse using a discarded plastic vaccine bottle as a makeshift funnel to transfer liquid between vials. Not ideal, obviously, but resourceful.
And then there’s storage. People will store all sorts of things in these bottles once the vaccine is used up. I’ve heard stories of people using them to store pills, vitamins, even small tools. It's a testament to their durability, I guess.
The biggest advantage of plastic, of course, is its weight. Glass is heavy, and heavy means expensive to ship. Plastic is lightweight, durable, and relatively inexpensive. The disadvantages? Well, permeability can be an issue, especially with certain solvents. And, let's be real, it's not as aesthetically pleasing as glass. But for a plastic vaccine bottle, functionality is paramount.
Customization is key. We can mold in different colors, add graduation marks, print logos… We even had one client who wanted a QR code printed directly onto the bottle for track-and-trace purposes. That was a challenge, but we figured it out.
Last month, that small boss in Shenzhen who makes smart home devices insisted on changing the interface to . He wanted it to look “modern.” He thought it would be a selling point. The result was… a disaster. The connector was too bulky for the bottle neck, and we couldn’t get a reliable seal. We lost weeks of production time, and he ended up having to go back to the old design. A complete waste of time and money.
That’s a perfect example of why you need to listen to the engineers. They’re the ones who know what’s actually feasible. Design is important, sure, but it has to be grounded in reality.
Anyway, I think that’s enough rambling for one day.
| Parameter | Importance (1-10) | Typical Range | Potential Issues |
|---|---|---|---|
| Neck Finish Diameter | 9 | 13mm - 18mm | Leakage, Compatibility with closures |
| Wall Thickness | 8 | 0.25mm - 0.5mm | Buckling, Cracking |
| Shoulder Radius | 7 | 5mm - 10mm | Stress Concentration, Cracking |
| Material Density | 6 | 0.90 - 0.92 g/cm³ | Weight Variations, Permeability |
| Closure Torque | 8 | 5-8 Nm | Over-tightening, Under-tightening |
| Bottle Volume | 7 | 5ml - 20ml | Filling Speed, Wastage |
The most common sterilization methods include gamma irradiation, ethylene oxide (EtO) sterilization, and autoclaving. However, not all plastics can withstand autoclaving, so it's crucial to choose a material compatible with the intended sterilization process. Gamma irradiation is preferred as it leaves no residue, but it can sometimes affect the polymer’s properties. EtO is effective but requires careful degassing to remove any residual gas. Proper validation of the sterilization process is absolutely essential to ensure the bottle remains sterile after processing.
Temperature fluctuations can significantly impact the plastic’s mechanical properties and permeability. Extreme heat can cause softening or deformation, while cold temperatures can make the plastic brittle and prone to cracking. Extended exposure to high temperatures can also accelerate degradation. We always recommend storing plastic vaccine bottles within a specified temperature range, usually between 2°C and 8°C, and avoiding direct sunlight or proximity to heat sources. Proper insulated packaging is essential for maintaining consistent temperatures during transport.
We conduct rigorous particulate matter testing using light obscuration methods and microscopic inspection. Bottles are rinsed with water for injection (WFI), and the resulting liquid is analyzed for the presence of particles of various sizes. We follow USP standards for particulate matter in injectable products. We also perform visual inspections under controlled lighting to identify any visible defects or contaminants. It’s a pain, but it's crucial for ensuring product safety.
Many plastic vaccine bottles are made from polypropylene (PP), which is technically recyclable, but the actual recycling rate is unfortunately low. Contamination from residual vaccine and the complexity of separating different plastic types hinder the recycling process. We’re actively exploring the use of post-consumer recycled (PCR) PP, but it requires careful validation to ensure it meets quality and safety standards. Reducing plastic consumption through optimized bottle design and promoting responsible disposal practices are also key environmental considerations.
The shelf life of an unopened plastic vaccine bottle depends on the material, storage conditions, and the specific vaccine it’s intended to hold. Typically, it’s around 2-3 years, but this is determined through extensive stability testing. We conduct accelerated aging studies at elevated temperatures and humidity levels to predict the bottle’s performance over time. The data is then used to establish a scientifically justified expiration date, ensuring the bottle maintains its integrity and doesn’t leach any harmful substances into the vaccine.
Ensuring compatibility is critical. We perform extractables and leachables studies, where we expose the plastic to the vaccine formulation and analyze for any substances that migrate from the plastic into the vaccine. We also assess the impact of the vaccine on the plastic’s physical and chemical properties. We follow guidelines set by regulatory bodies like the FDA and EMA. It's a long process, but absolutely necessary to prevent any adverse reactions or loss of vaccine potency.
Ultimately, plastic vaccine bottles are more than just containers; they’re a crucial link in the global healthcare supply chain. Getting the material right, the design right, the testing right... it all comes down to ensuring the vaccine reaches the patient safely and effectively. It’s a complex process, filled with potential pitfalls, but the stakes are too high to compromise.
And in the end, whether this thing works or not, the worker will know the moment he tightens the screw. If it feels solid, if it seals right, if it looks right... that's when you know you've got a good bottle. That’s the gut check that all the fancy tests and simulations can’t replace. You can visit our website at www.wkpacking.com to explore our range of plastic vaccine bottle solutions.